Muscle Releases Alpha-Sarcoglycan Positive Extracellular Vesicles Carrying miRNAs in the Bloodstream
- PMID: 25955720
- PMCID: PMC4425492
- DOI: 10.1371/journal.pone.0125094
Muscle Releases Alpha-Sarcoglycan Positive Extracellular Vesicles Carrying miRNAs in the Bloodstream
Abstract
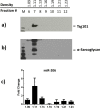
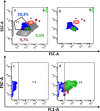
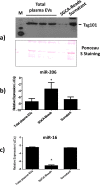
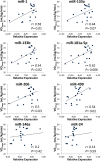
In the past few years, skeletal muscle has emerged as an important secretory organ producing soluble factors, called myokines, that exert either autocrine, paracrine or endocrine effects. Moreover, recent studies have shown that muscle releases microRNAs into the bloodstream in response to physical exercise. These microRNAs affect target cells, such as hormones and cytokines. The mechanisms underlying microRNA secretion are poorly characterized at present. Here, we investigated whether muscle tissue releases extracellular vesicles (EVs), which carry microRNAs in the bloodstream under physiological conditions such as physical exercise. Using density gradient separation of plasma from sedentary and physically fit young men we found EVs positive for TSG101 and alpha-sarcoglycan (SGCA), and enriched for miR-206. Cytometric analysis showed that the SGCA+ EVs account for 1-5% of the total and that 60-65% of these EVs were also positive for the exosomal marker CD81. Furthermore, the SGCA-immuno captured sub-population of EVs exhibited higher levels of the miR-206/miR16 ratio compared to total plasma EVs. Finally, a significant positive correlation was found between the aerobic fitness and muscle-specific miRNAs and EV miR-133b and -181a-5p were significantly up-regulated after acute exercise. Thus, our study proposes EVs as a novel means of muscle communication potentially involved in muscle remodeling and homeostasis.
Conflict of interest statement
Figures

References
-
- Goldstein M. Humoral nature of the hypoglycemic factor of muscular work. Diabetes. 1961;10: 232–234. - PubMed
-
- Pedersen B, Steensberg A, Fischer C, Keller C, Keller P, Plomgaard P, et al. Searching for the exercise factor: is IL-6 a candidate? Journal of muscle research and cell motility. 2003;24: 113–119. - PubMed
-
- Bortoluzzi S, Scannapieco P, Cestaro A, Danieli GA, Schiaffino S. Computational reconstruction of the human skeletal muscle secretome. Proteins. 2006;62: 776–792. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

