Topical, Aqueous, Clear Cyclosporine Formulation Design for Anterior and Posterior Ocular Delivery
- PMID: 25964868
- PMCID: PMC4418434
- DOI: 10.1167/tvst.4.3.1
Topical, Aqueous, Clear Cyclosporine Formulation Design for Anterior and Posterior Ocular Delivery
Abstract
Purpose: The main objective of this study was to optimize cyclosporine (CsA) nanomicellar solution and study in vivo ocular CsA tissue distribution with a topical drop.
Methods: An optimized blend of hydrogenated castor oil-40 and octoxynol-40 was prepared to entrap CsA within nanomicelles. In vivo studies were conducted in New Zealand White albino rabbits with topical drop instillation.
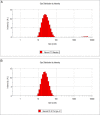
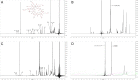
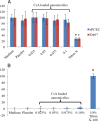
Results: Average size of CsA-loaded nanomicelles was approximately 22.4 nm. Ocular tissue CsA quantification with single and multiple dosing revealed that CsA levels followed as cornea → iris-ciliary body → aqueous humor → lens. Cyclosporine levels were also found to be in the following order: conjunctiva → sclera → retina/choroid → vitreous humor. High CsA level was detected in retina/choroid (53.7 ng/g tissue).
Conclusions: Ocular tissue CsA distribution studies revealed high CsA concentrations in anterior ocular tissues. Moreover, it appears that nanomicelles are transported through a conjunctival-scleral pathway and deliver CsA to the retina/choroid. Results suggest polymeric blend to be a safe carrier for anterior and posterior ocular tissues.
Translational relevance: This study has significant translational relevance, disclosing results that suggest that aqueous nanomicellar approach can provide high corneal and conjunctival CsA concentrations. Aqueous nanomicelles can deliver high drug concentrations not only to anterior but also to back of the eye tissues, including retina. This article provides a platform for noninvasive back of the eye drug delivery with topical eye drops. Aqueous CsA nanomicelles have no perceptible toxicity such as cell membrane damage or cytotoxicity to corneal and retinal pigment epithelial cells. Clear aqueous nanomicellar solution can be translated to human conditions for keratoconjunctivitis sicca and other anti-inflammatory conditions.
Keywords: back of the eye; cyclosporine; drops; drug delivery; dry eye; formulation; nanomicelles; posterior; rabbits; retina/choroid; sclera; topical.
Figures

Similar articles
-
Ocular Distribution of Cyclosporine Following Topical Administration of OTX-101 in New Zealand White Rabbits.J Ocul Pharmacol Ther. 2019 Sep;35(7):395-402. doi: 10.1089/jop.2018.0106. Epub 2019 Aug 6. J Ocul Pharmacol Ther. 2019. PMID: 31355703 Free PMC article.
-
Topical delivery of aqueous micellar resolvin E1 analog (RX-10045).Int J Pharm. 2016 Feb 10;498(1-2):326-34. doi: 10.1016/j.ijpharm.2015.12.037. Epub 2015 Dec 17. Int J Pharm. 2016. PMID: 26706439 Free PMC article.
-
Nanomicellar Topical Aqueous Drop Formulation of Rapamycin for Back-of-the-Eye Delivery.AAPS PharmSciTech. 2015 Jun;16(3):610-22. doi: 10.1208/s12249-014-0244-2. Epub 2014 Nov 26. AAPS PharmSciTech. 2015. PMID: 25425389 Free PMC article.
-
A Review of Topical Cyclosporine A Formulations-A Disease-Modifying Agent for Keratoconjunctivitis Sicca.Clin Ophthalmol. 2020 Feb 20;14:481-489. doi: 10.2147/OPTH.S228070. eCollection 2020. Clin Ophthalmol. 2020. PMID: 32109984 Free PMC article. Review.
-
Ocular Pharmacokinetics of a Topical Ophthalmic Nanomicellar Solution of Cyclosporine (Cequa®) for Dry Eye Disease.Pharm Res. 2019 Jan 7;36(2):36. doi: 10.1007/s11095-018-2556-5. Pharm Res. 2019. PMID: 30617777 Review.
Cited by
-
Recent perspectives on the delivery of biologics to back of the eye.Expert Opin Drug Deliv. 2017 May;14(5):631-645. doi: 10.1080/17425247.2016.1227783. Epub 2016 Sep 6. Expert Opin Drug Deliv. 2017. PMID: 27573097 Free PMC article. Review.
-
The Emerging Role of Topical Ocular Drugs to Target the Posterior Eye.Ophthalmol Ther. 2021 Sep;10(3):465-494. doi: 10.1007/s40123-021-00365-y. Epub 2021 Jul 4. Ophthalmol Ther. 2021. PMID: 34218424 Free PMC article. Review.
-
Managing Dry Eye Disease and Facilitating Realistic Patient Expectations: A Review and Appraisal of Current Therapies.Clin Ophthalmol. 2020 Jan 14;14:119-126. doi: 10.2147/OPTH.S228838. eCollection 2020. Clin Ophthalmol. 2020. PMID: 32021076 Free PMC article. Review.
-
Clinical Efficacy and Safety of Preservative Free Cyclosporine in Dry Eye Disease.Clin Ophthalmol. 2025 Mar 31;19:1149-1154. doi: 10.2147/OPTH.S505641. eCollection 2025. Clin Ophthalmol. 2025. PMID: 40191005 Free PMC article. Clinical Trial.
-
Ocular delivery of proteins and peptides: Challenges and novel formulation approaches.Adv Drug Deliv Rev. 2018 Feb 15;126:67-95. doi: 10.1016/j.addr.2018.01.008. Epub 2018 Jan 13. Adv Drug Deliv Rev. 2018. PMID: 29339145 Free PMC article. Review.
References
-
- Mondon K,, Zeisser-Labouebe M, Gurny R,, et al. Novel cyclosporin A formulations using MPEG-hexyl-substituted polylactide micelles: a suitability study. Eur J Pharm Biopharm. 2011; 77: 56–65. - PubMed
-
- Matsuda S, Koyasu S.Mechanisms of action of cyclosporine. Immunopharmacology. 2000; 47: 119–125. - PubMed
-
- Whitcup SM, Chan CC,, Luyo DA, et al. Topical cyclosporine inhibits mast cell-mediated conjunctivitis. Invest Ophthalmol Vis Sci. 1996; 37: 2686–2693. - PubMed
-
- Kunert KS,, Tisdale AS, Gipson IK.Goblet cell numbers and epithelial proliferation in the conjunctiva of patients with dry eye syndrome treated with cyclosporine. Arch Ophthalmol. 2002; 120: 330–337. - PubMed
-
- Gunduz K, Ozdemir O.Topical cyclosporin treatment of keratoconjunctivitis sicca in secondary Sjogren's syndrome. Acta Ophthalmol (Copenh). 1994; 72: 438–442. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

