Edin Expression in the Fat Body Is Required in the Defense Against Parasitic Wasps in Drosophila melanogaster
- PMID: 25965263
- PMCID: PMC4429011
- DOI: 10.1371/journal.ppat.1004895
Edin Expression in the Fat Body Is Required in the Defense Against Parasitic Wasps in Drosophila melanogaster
Abstract
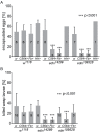
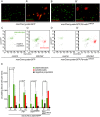
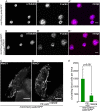
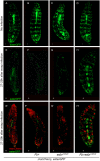
The cellular immune response against parasitoid wasps in Drosophila involves the activation, mobilization, proliferation and differentiation of different blood cell types. Here, we have assessed the role of Edin (elevated during infection) in the immune response against the parasitoid wasp Leptopilina boulardi in Drosophila melanogaster larvae. The expression of edin was induced within hours after a wasp infection in larval fat bodies. Using tissue-specific RNAi, we show that Edin is an important determinant of the encapsulation response. Although edin expression in the fat body was required for the larvae to mount a normal encapsulation response, it was dispensable in hemocytes. Edin expression in the fat body was not required for lamellocyte differentiation, but it was needed for the increase in plasmatocyte numbers and for the release of sessile hemocytes into the hemolymph. We conclude that edin expression in the fat body affects the outcome of a wasp infection by regulating the increase of plasmatocyte numbers and the mobilization of sessile hemocytes in Drosophila larvae.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Benassi V, Coustau C, Carton Y. Insect immunity: A genetic factor (hrtp) is essential for antibacterial peptide expression in drosophila after infection by parasitoid wasps. Arch Insect Biochem Physiol 2000; 43(2): 64–71. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

