Hepatitis C Virus NS3 Mediated Microglial Inflammation via TLR2/TLR6 MyD88/NF-κB Pathway and Toll Like Receptor Ligand Treatment Furnished Immune Tolerance
- PMID: 25965265
- PMCID: PMC4428696
- DOI: 10.1371/journal.pone.0125419
Hepatitis C Virus NS3 Mediated Microglial Inflammation via TLR2/TLR6 MyD88/NF-κB Pathway and Toll Like Receptor Ligand Treatment Furnished Immune Tolerance
Abstract
Background: Recent evidence suggests the neurotrophic potential of hepatitis C virus (HCV). HCV NS3 protein is one of the potent antigens of this virus mediating inflammatory response in different cell types. Microglia being the immune surveillance cells in the central nervous system (CNS), the inflammatory potential of NS3 on microglia was studied. Role of toll like receptor (TLR) ligands Pam2CSK3 and Pam3CSK4 in controlling the NS3 mediated microglial inflammation was studied using microglial cell line CHME3.
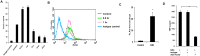
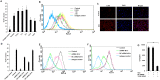
Methods: IL (Interleukin)-8, IL-6, TNF-α (Tumor nicrosis factor alpha) and IL-1β gene expressions were measured by semi quantitative RT-PCR (reverse transcription-PCR). ELISA was performed to detect IL-8, IL-6, TNF-α, IL-1β and IL-10 secretion. FACS (Flourescent activated cell sorting) was performed to quantify TLR1, TLR2, TLR6, MyD88 (Myeloid differntiation factor 88), IkB-α (I kappaB alpha) and pNF-κB (phosphorylated nuclear factor kappaB) expression. Immunofluorescence staining was performed for MyD88, TLR6 and NF-κB (Nuclear factor kappaB). Student's t-test or One way analysis of variance with Bonferoni post hoc test was performed and p < 0.05 was considered significant.
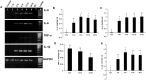
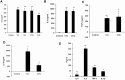
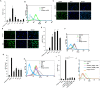
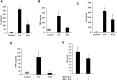
Results: Microglia responded to NS3 by secreting IL-8, IL-6, TNF-α and IL-1β via TLR2 or TLR6 mediated MyD88/NF-κB pathway. Transcription factor NF-κB was involved in activating the cytokine gene expression and the resultant inflammatory response was controlled by NF-κB inhibitor, Ro106-9920, which is known to down regulate pro-inflammatory cytokine secretion. Activation of the microglia by TLR agonists Pam3CSK4 and Pam2CSK4 induced immune tolerance against NS3. TLR ligand treatment significantly down regulated pro-inflammatory cytokine secretion in the microglia. IL-10 secretion was suggested as the possible mechanism by which TLR agonists induced immune tolerance. NS3 as such was not capable of self-inducing immune tolerance in microglia.
Conclusion: In conclusion, NS3 protein was capable of activating microglia and the inflammatory response could be controlled via blocking the transcription factor NF-κB, or by treating the microglia with TLR ligands which likely function via secreting anti-inflammatory cytokines such as IL-10. This can have therapeutic potential in controlling HCV mediated neuroinflammation.
Conflict of interest statement
Figures




Similar articles
-
Hepatitis C virus core and NS3 antigens induced conjunctival inflammation via toll-like receptor-mediated signaling.Mol Vis. 2014 Sep 25;20:1388-97. eCollection 2014. Mol Vis. 2014. PMID: 25352745 Free PMC article.
-
HCV core and NS3 proteins mediate toll like receptor induced innate immune response in corneal epithelium.Exp Eye Res. 2014 Nov;128:117-28. doi: 10.1016/j.exer.2014.09.011. Epub 2014 Oct 1. Exp Eye Res. 2014. PMID: 25280963
-
Anti-inflammatory protein TSG-6 secreted by bone marrow mesenchymal stem cells attenuates neuropathic pain by inhibiting the TLR2/MyD88/NF-κB signaling pathway in spinal microglia.J Neuroinflammation. 2020 May 11;17(1):154. doi: 10.1186/s12974-020-1731-x. J Neuroinflammation. 2020. PMID: 32393298 Free PMC article.
-
Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway.J Neuroinflammation. 2019 Jul 18;16(1):148. doi: 10.1186/s12974-019-1538-9. J Neuroinflammation. 2019. PMID: 31319868 Free PMC article. Review.
-
Effects of Toll-like receptor 1 and 2 agonist Pam3CSK4 on uveal melanocytes and relevant experimental mouse model.Exp Eye Res. 2024 Feb;239:109749. doi: 10.1016/j.exer.2023.109749. Epub 2023 Dec 17. Exp Eye Res. 2024. PMID: 38113956 Review.
Cited by
-
TLR3-Dependent Activation of TLR2 Endogenous Ligands via the MyD88 Signaling Pathway Augments the Innate Immune Response.Cells. 2020 Aug 17;9(8):1910. doi: 10.3390/cells9081910. Cells. 2020. PMID: 32824595 Free PMC article.
-
Cell membrane-bound toll-like receptor-1/2/4/6 monomers and -2 heterodimer inhibit enterovirus 71 replication by activating the antiviral innate response.Front Immunol. 2023 May 3;14:1187035. doi: 10.3389/fimmu.2023.1187035. eCollection 2023. Front Immunol. 2023. PMID: 37207203 Free PMC article.
-
From viruses to cancer: exploring the role of the hepatitis C virus NS3 protein in carcinogenesis.Infect Agent Cancer. 2024 Aug 27;19(1):40. doi: 10.1186/s13027-024-00606-2. Infect Agent Cancer. 2024. PMID: 39192306 Free PMC article. Review.
-
Mitochondrial-Directed Antioxidant Reduces Microglial-Induced Inflammation in Murine In Vitro Model of TC-83 Infection.Viruses. 2018 Nov 2;10(11):606. doi: 10.3390/v10110606. Viruses. 2018. PMID: 30400156 Free PMC article.
-
The human microglial HMC3 cell line: where do we stand? A systematic literature review.J Neuroinflammation. 2018 Sep 10;15(1):259. doi: 10.1186/s12974-018-1288-0. J Neuroinflammation. 2018. PMID: 30200996 Free PMC article. Review.
References
-
- Howard CR. Hepatitis C virus: clades and properties. J Gastroentrol Hepatol. 2002; 17: S468–470. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

