Crossmodal integration improves sensory detection thresholds in the ferret
- PMID: 25970327
- PMCID: PMC4430165
- DOI: 10.1371/journal.pone.0124952
Crossmodal integration improves sensory detection thresholds in the ferret
Abstract
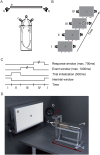
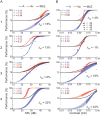
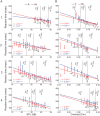
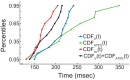
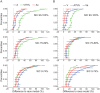
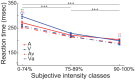
During the last two decades ferrets (Mustela putorius) have been established as a highly efficient animal model in different fields in neuroscience. Here we asked whether ferrets integrate sensory information according to the same principles established for other species. Since only few methods and protocols are available for behaving ferrets we developed a head-free, body-restrained approach allowing a standardized stimulation position and the utilization of the ferret's natural response behavior. We established a behavioral paradigm to test audiovisual integration in the ferret. Animals had to detect a brief auditory and/or visual stimulus presented either left or right from their midline. We first determined detection thresholds for auditory amplitude and visual contrast. In a second step, we combined both modalities and compared psychometric fits and the reaction times between all conditions. We employed Maximum Likelihood Estimation (MLE) to model bimodal psychometric curves and to investigate whether ferrets integrate modalities in an optimal manner. Furthermore, to test for a redundant signal effect we pooled the reaction times of all animals to calculate a race model. We observed that bimodal detection thresholds were reduced and reaction times were faster in the bimodal compared to unimodal conditions. The race model and MLE modeling showed that ferrets integrate modalities in a statistically optimal fashion. Taken together, the data indicate that principles of multisensory integration previously demonstrated in other species also apply to crossmodal processing in the ferret.
Conflict of interest statement
Figures

Similar articles
-
Primary auditory cortex in the ferret (Mustela putorius): neural response properties and topographic organization.Brain Res. 1988 Mar 8;443(1-2):281-94. doi: 10.1016/0006-8993(88)91622-8. Brain Res. 1988. PMID: 3359271
-
Representation of the cochlea in primary auditory cortex of the ferret (Mustela putorius).Hear Res. 1986;24(2):111-5. doi: 10.1016/0378-5955(86)90054-7. Hear Res. 1986. PMID: 3771373
-
Behavioural benefits of multisensory processing in ferrets.Eur J Neurosci. 2017 Jan;45(2):278-289. doi: 10.1111/ejn.13440. Epub 2016 Nov 3. Eur J Neurosci. 2017. PMID: 27740711 Free PMC article.
-
Auditory influences on non-auditory cortices.Hear Res. 2009 Dec;258(1-2):64-71. doi: 10.1016/j.heares.2009.03.005. Epub 2009 Mar 20. Hear Res. 2009. PMID: 19303926 Free PMC article. Review.
-
Visual influences on ferret auditory cortex.Hear Res. 2009 Dec;258(1-2):55-63. doi: 10.1016/j.heares.2009.06.017. Epub 2009 Jul 10. Hear Res. 2009. PMID: 19595754 Free PMC article. Review.
Cited by
-
Cross-Modal Stochastic Resonance as a Universal Principle to Enhance Sensory Processing.Front Neurosci. 2018 Aug 21;12:578. doi: 10.3389/fnins.2018.00578. eCollection 2018. Front Neurosci. 2018. PMID: 30186104 Free PMC article. No abstract available.
-
Visual Motion and Form Integration in the Behaving Ferret.eNeuro. 2019 Aug 20;6(4):ENEURO.0228-19.2019. doi: 10.1523/ENEURO.0228-19.2019. Print 2019 Jul/Aug. eNeuro. 2019. PMID: 31371456 Free PMC article.
-
Differential Rapid Plasticity in Auditory and Visual Responses in the Primarily Multisensory Orbitofrontal Cortex.eNeuro. 2020 Jun 12;7(3):ENEURO.0061-20.2020. doi: 10.1523/ENEURO.0061-20.2020. Print 2020 May/Jun. eNeuro. 2020. PMID: 32424057 Free PMC article.
-
Audiovisual Integration Enhances Stimulus Detection Performance in Mice.Front Behav Neurosci. 2018 Oct 4;12:231. doi: 10.3389/fnbeh.2018.00231. eCollection 2018. Front Behav Neurosci. 2018. PMID: 30337861 Free PMC article.
-
Cross-Talk of Low-Level Sensory and High-Level Cognitive Processing: Development, Mechanisms, and Relevance for Cross-Modal Abilities of the Brain.Front Neurorobot. 2020 Feb 14;14:7. doi: 10.3389/fnbot.2020.00007. eCollection 2020. Front Neurorobot. 2020. PMID: 32116637 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

