Intralesional administration of L19-IL2/L19-TNF in stage III or stage IVM1a melanoma patients: results of a phase II study
- PMID: 25971540
- PMCID: PMC11028725
- DOI: 10.1007/s00262-015-1704-6
Intralesional administration of L19-IL2/L19-TNF in stage III or stage IVM1a melanoma patients: results of a phase II study
Abstract
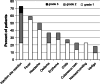
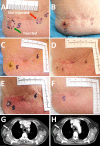
The intratumoral injection of cytokines, in particular IL2, has shown promise for cutaneous melanoma patients with unresectable disease or continuous recurrence despite surgery. We recently reported that the intralesional injection of L19-IL2, an immunocytokine combining IL2 and the human monoclonal antibody fragment L19, resulted in efficient regional control of disease progression, increased time to distant metastasis and evidence of effect on circulating immune cell populations. We have also shown in preclinical models of cancer a remarkable synergistic effect of the combination of L19-IL2 with L19-TNF, a second clinical-stage immunocytokine, based on the same L19 antibody fused to TNF. Here, we describe the results of a phase II clinical trial based on the intralesional administration of L19-IL2 and L19-TNF in patients with stage IIIC and IVM1a metastatic melanoma, who were not candidate to surgery. In 20 efficacy-evaluable patients, 32 melanoma lesions exhibited complete responses upon intralesional administration of the two products, with mild side effects mainly limited to injection site reactions. Importantly, we observed complete responses in 7/13 (53.8 %) non-injected lesions (4 cutaneous, 3 lymph nodes), indicating a systemic activity of the intralesional immunostimulatory treatment. The intralesional administration of L19-IL2 and L19-TNF represents a simple and effective method for the local control of inoperable melanoma lesions, with a potential to eradicate them or make them suitable for a facile surgical removal of the residual mass.
Figures


Similar articles
-
Intralesional treatment of stage III metastatic melanoma patients with L19-IL2 results in sustained clinical and systemic immunologic responses.Cancer Immunol Res. 2014 Jul;2(7):668-78. doi: 10.1158/2326-6066.CIR-13-0206. Epub 2014 Apr 4. Cancer Immunol Res. 2014. PMID: 24906352 Clinical Trial.
-
The immunocytokine L19-IL2 eradicates cancer when used in combination with CTLA-4 blockade or with L19-TNF.J Invest Dermatol. 2013 Mar;133(3):751-758. doi: 10.1038/jid.2012.376. Epub 2012 Oct 25. J Invest Dermatol. 2013. PMID: 23096716
-
A dose-escalation and signal-generating study of the immunocytokine L19-IL2 in combination with dacarbazine for the therapy of patients with metastatic melanoma.Clin Cancer Res. 2011 Dec 15;17(24):7732-42. doi: 10.1158/1078-0432.CCR-11-1203. Epub 2011 Oct 25. Clin Cancer Res. 2011. PMID: 22028492 Clinical Trial.
-
Pharmacokinetic drug evaluation of talimogene laherparepvec for the treatment of advanced melanoma.Expert Opin Drug Metab Toxicol. 2018 Apr;14(4):469-473. doi: 10.1080/17425255.2018.1455825. Epub 2018 Mar 23. Expert Opin Drug Metab Toxicol. 2018. PMID: 29557682 Review.
-
Practical clinical guide on the use of talimogene laherparepvec monotherapy in patients with unresectable melanoma in Europe.Eur J Dermatol. 2018 Dec 1;28(6):736-749. doi: 10.1684/ejd.2018.3447. Eur J Dermatol. 2018. PMID: 30698145 Review.
Cited by
-
Effects of interleukin-2 in immunostimulation and immunosuppression.J Exp Med. 2020 Jan 6;217(1):e20191247. doi: 10.1084/jem.20191247. J Exp Med. 2020. PMID: 31611250 Free PMC article. Review.
-
Trial watch: intratumoral immunotherapy.Oncoimmunology. 2021 Oct 15;10(1):1984677. doi: 10.1080/2162402X.2021.1984677. eCollection 2021. Oncoimmunology. 2021. PMID: 34676147 Free PMC article. Review.
-
A novel strategy to generate immunocytokines with activity-on-demand using small molecule inhibitors.EMBO Mol Med. 2024 Apr;16(4):904-926. doi: 10.1038/s44321-024-00034-0. Epub 2024 Mar 6. EMBO Mol Med. 2024. PMID: 38448543 Free PMC article.
-
Evolution of phage display libraries for therapeutic antibody discovery.MAbs. 2023 Jan-Dec;15(1):2213793. doi: 10.1080/19420862.2023.2213793. MAbs. 2023. PMID: 37222232 Free PMC article. Review.
-
Enhanced Therapeutic Activity of Non-Internalizing Small-Molecule-Drug Conjugates Targeting Carbonic Anhydrase IX in Combination with Targeted Interleukin-2.Clin Cancer Res. 2018 Aug 1;24(15):3656-3667. doi: 10.1158/1078-0432.CCR-17-3457. Epub 2018 Apr 24. Clin Cancer Res. 2018. PMID: 29691298 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

