Fungal traits that drive ecosystem dynamics on land
- PMID: 25971588
- PMCID: PMC4429240
- DOI: 10.1128/MMBR.00001-15
Fungal traits that drive ecosystem dynamics on land
Abstract
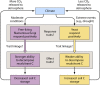
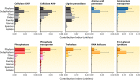
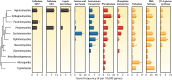
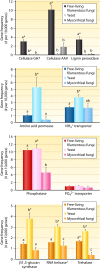
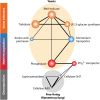
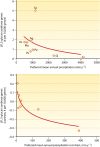
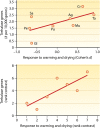
Fungi contribute extensively to a wide range of ecosystem processes, including decomposition of organic carbon, deposition of recalcitrant carbon, and transformations of nitrogen and phosphorus. In this review, we discuss the current knowledge about physiological and morphological traits of fungi that directly influence these processes, and we describe the functional genes that encode these traits. In addition, we synthesize information from 157 whole fungal genomes in order to determine relationships among selected functional genes within fungal taxa. Ecosystem-related traits varied most at relatively coarse taxonomic levels. For example, we found that the maximum amount of variance for traits associated with carbon mineralization, nitrogen and phosphorus cycling, and stress tolerance could be explained at the levels of order to phylum. Moreover, suites of traits tended to co-occur within taxa. Specifically, the genetic capacities for traits that improve stress tolerance-β-glucan synthesis, trehalose production, and cold-induced RNA helicases-were positively related to one another, and they were more evident in yeasts. Traits that regulate the decomposition of complex organic matter-lignin peroxidases, cellobiohydrolases, and crystalline cellulases-were also positively related, but they were more strongly associated with free-living filamentous fungi. Altogether, these relationships provide evidence for two functional groups: stress tolerators, which may contribute to soil carbon accumulation via the production of recalcitrant compounds; and decomposers, which may reduce soil carbon stocks. It is possible that ecosystem functions, such as soil carbon storage, may be mediated by shifts in the fungal community between stress tolerators and decomposers in response to environmental changes, such as drought and warming.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures
References
-
- Dighton J. 2003. Fungi in ecosystem processes, vol 17 Marcel Dekker, New York, NY.
-
- Chapin FS, Matson PA, Vitousek PM, Chapin MC. 2011. Principles of terrestrial ecosystem ecology, 2nd ed Springer, New York, NY.
-
- Kogel-Knabner I. 2002. The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter. Soil Biol Biochem 34:139–162. doi: 10.1016/S0038-0717(01)00158-4. - DOI
-
- Six J, Frey SD, Thiet RK, Batten KM. 2006. Bacterial and fungal contributions to carbon sequestration in agroecosystems. Soil Sci Soc Am J 70:555–569. doi: 10.2136/sssaj2004.0347. - DOI

