Chitosan nanoparticles loaded with the antimicrobial peptide temporin B exert a long-term antibacterial activity in vitro against clinical isolates of Staphylococcus epidermidis
- PMID: 25972852
- PMCID: PMC4412066
- DOI: 10.3389/fmicb.2015.00372
Chitosan nanoparticles loaded with the antimicrobial peptide temporin B exert a long-term antibacterial activity in vitro against clinical isolates of Staphylococcus epidermidis
Abstract
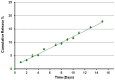
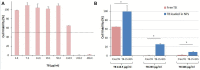
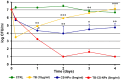
Nowadays, the alarming rise in multidrug-resistant microorganisms urgently demands for suitable alternatives to current antibiotics. In this regard, antimicrobial peptides (AMPs) have received growing interest due to their broad spectrum of activities, potent antimicrobial properties, unique mechanisms of action, and low tendency to induce resistance. However, their pharmaceutical development is hampered by potential toxicity, relatively low stability and manufacturing costs. In the present study, we tested the hypothesis that the encapsulation of the frog-skin derived AMP temporin B (TB) into chitosan nanoparticles (CS-NPs) could increase peptide's antibacterial activity, while reducing its toxic potential. TB-loaded CS-NPs with good dimensional features were prepared, based on the ionotropic gelation between CS and sodium tripolyphosphate. The encapsulation efficiency of TB in the formulation was up to 75%. Release kinetic studies highlighted a linear release of the peptide from the nanocarrier, in the adopted experimental conditions. Interestingly, the encapsulation of TB in CS-NPs demonstrated to reduce significantly the peptide's cytotoxicity against mammalian cells. Additionally, the nanocarrier evidenced a sustained antibacterial action against various strains of Staphylococcus epidermidis for at least 4 days, with up to 4-log reduction in the number of viable bacteria compared to plain CS-NPs at the end of the observational period. Of note, the antimicrobial evaluation tests demonstrated that while the intrinsic antimicrobial activity of CS ensured a "burst" effect, the gradual release of TB further reduced the viable bacterial count, preventing the regrowth of the residual cells and ensuring a long-lasting antibacterial effect. The developed nanocarrier is eligible for the administration of several AMPs of therapeutic interest with physical-chemical characteristics analog to those of TB.
Keywords: Staphylococcus epidermidis; antimicrobial peptides; chitosan; delivery system; nanoparticles; temporin B.
Figures

References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

