Activation of natriuretic peptides and the sympathetic nervous system following Roux-en-Y gastric bypass is associated with gonadal adipose tissues browning
- PMID: 25973390
- PMCID: PMC4421080
- DOI: 10.1016/j.molmet.2015.02.006
Activation of natriuretic peptides and the sympathetic nervous system following Roux-en-Y gastric bypass is associated with gonadal adipose tissues browning
Abstract
Objective: Roux-en-Y gastric bypass (RYGB) is an effective method of weight loss and remediation of type-2 diabetes; however, the mechanisms leading to these improvements are unclear. Additionally, adipocytes within white adipose tissue (WAT) depots can manifest characteristics of brown adipocytes. These 'BRITE/beige' adipocytes express uncoupling protein 1 (UCP1) and are associated with improvements in glucose homeostasis and protection from obesity. Interestingly, atrial and B-type natriuretic peptides (NPs) promote BRITE/beige adipocyte enrichment of WAT depots, an effect known as "browning." Here, we investigate the effect of RYGB surgery on NP, NP receptors, and browning in the gonadal adipose tissues of female mice. We propose that such changes may lead to improvements in metabolic homeostasis commonly observed following RYGB.
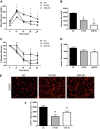
Methods: Wild type, female, C57/Bl6 mice were fed a 60% fat diet ad libitum for six months. Mice were divided into three groups: Sham operated (SO), Roux-en-Y gastric bypass (RYGB), and Weight matched, sham operated (WM-SO). Mice were sacrificed six weeks following surgery and evaluated for differences in body weight, glucose homeostasis, adipocyte morphology, and adipose tissue gene expression.
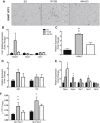
Results: RYGB and calorie restriction induced similar weight loss and improved glucose metabolism without decreasing food intake. β3-adrenergic receptor expression increased in gonadal adipose tissue, in addition to Nppb (BNP), and NP receptors, Npr1, and Npr2. The ratio of Npr1:Npr3 and Npr2:Npr3 increased in RYGB, but not WM-SO groups. Ucp1 protein and mRNA, as well as additional markers of BRITE/beige adipose tissue and lipolytic genes increased in RYGB mice to a greater extent than calorie-restricted mice.
Conclusions: Upregulation of Nppb, Npr1, Npr2, and β3-adrenergic receptors in gonadal adipose tissue following RYGB was associated with increased markers of browning. This browning of gonadal adipose tissue may underpin the positive effect of RYGB on metabolic parameters and may in part be mediated through upregulation of natriuretic peptides.
Keywords: Atrial natriuretic peptide (ANP); B-type natriuretic peptide (BNP); Browning; High fat diet (HFD); Natriuretic peptide receptor; Roux-en-Y gastric bypass (RYGB).
Figures

References
-
- Sanchis P., Frances C., Nicolau J., Rivera R., Fortuny R., Julian X. Cardiovascular risk profile in Mediterranean patients submitted to bariatric surgery and intensive lifestyle intervention: impact of both interventions after 1 year of follow-up. Obesity Surgery. 2014 - PubMed
-
- Bielefeldt K. Bariatric surgery versus intensive medical therapy for diabetes. The New England Journal of Medicine. 2014;371(7):681–682. - PubMed
-
- Surgery, A.S.f.M.a.B., Bariatric Surgery Procedures.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

