Molecular-interaction and signaling profiles of AM3677, a novel covalent agonist selective for the cannabinoid 1 receptor
- PMID: 25978068
- PMCID: PMC4804703
- DOI: 10.1021/acschemneuro.5b00090
Molecular-interaction and signaling profiles of AM3677, a novel covalent agonist selective for the cannabinoid 1 receptor
Abstract
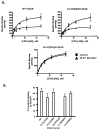
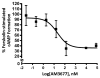
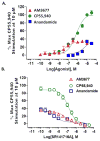
The cannabinoid 1 receptor (CB1R) is one of the most abundant G protein-coupled receptors (GPCRs) in the central nervous system. CB1R involvement in multiple physiological processes, especially neurotransmitter release and synaptic function, has made this GPCR a prime drug discovery target, and pharmacological CB1R activation has been demonstrated to be a tenable therapeutic modality. Accordingly, the design and profiling of novel, drug-like CB1R modulators to inform the receptor's ligand-interaction landscape and molecular pharmacology constitute a prime contemporary research focus. For this purpose, we report utilization of AM3677, a designer endocannabinoid (anandamide) analogue derivatized with a reactive electrophilic isothiocyanate functionality, as a covalent, CB1R-selective chemical probe. The data demonstrate that reaction of AM3677 with a cysteine residue in transmembrane helix 6 of human CB1R (hCB1R), C6.47(355), is a key feature of AM3677's ligand-binding motif. Pharmacologically, AM3677 acts as a high-affinity, low-efficacy CB1R agonist that inhibits forskolin-stimulated cellular cAMP formation and stimulates CB1R coupling to G protein. AM3677 also induces CB1R endocytosis and irreversible receptor internalization. Computational docking suggests the importance of discrete hydrogen bonding and aromatic interactions as determinants of AM3677's topology within the ligand-binding pocket of active-state hCB1R. These results constitute the initial identification and characterization of a potent, high-affinity, hCB1R-selective covalent agonist with utility as a pharmacologically active, orthosteric-site probe for providing insight into structure-function correlates of ligand-induced CB1R activation and the molecular features of that activation by the native ligand, anandamide.
Keywords: 7-transmembrane receptor; Amino acid; G protein-coupled receptor; binding domain; central nervous system; chemical probe; cysteine; homology modeling; isothiocyanate; ligand-binding motif; receptor activation; signal transduction.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Mapping Cannabinoid 1 Receptor Allosteric Site(s): Critical Molecular Determinant and Signaling Profile of GAT100, a Novel, Potent, and Irreversibly Binding Probe.ACS Chem Neurosci. 2016 Jun 15;7(6):776-98. doi: 10.1021/acschemneuro.6b00041. Epub 2016 Apr 25. ACS Chem Neurosci. 2016. PMID: 27046127 Free PMC article.
-
Human Cannabinoid Receptor 2 Ligand-Interaction Motif: Transmembrane Helix 2 Cysteine, C2.59(89), as Determinant of Classical Cannabinoid Agonist Activity and Binding Pose.ACS Chem Neurosci. 2017 Jun 21;8(6):1338-1347. doi: 10.1021/acschemneuro.7b00003. Epub 2017 Mar 1. ACS Chem Neurosci. 2017. PMID: 28220706
-
Enantiospecific Allosteric Modulation of Cannabinoid 1 Receptor.ACS Chem Neurosci. 2017 Jun 21;8(6):1188-1203. doi: 10.1021/acschemneuro.6b00310. Epub 2017 Feb 7. ACS Chem Neurosci. 2017. PMID: 28103441
-
Leveraging allostery to improve G protein-coupled receptor (GPCR)-directed therapeutics: cannabinoid receptor 1 as discovery target.Expert Opin Drug Discov. 2016 Dec;11(12):1223-1237. doi: 10.1080/17460441.2016.1245289. Epub 2016 Oct 21. Expert Opin Drug Discov. 2016. PMID: 27712124 Free PMC article. Review.
-
Allosteric Modulation of Cannabinoid Receptor 1-Current Challenges and Future Opportunities.Int J Mol Sci. 2019 Nov 22;20(23):5874. doi: 10.3390/ijms20235874. Int J Mol Sci. 2019. PMID: 31771126 Free PMC article. Review.
Cited by
-
Allosteric and orthosteric pharmacology of cannabidiol and cannabidiol-dimethylheptyl at the type 1 and type 2 cannabinoid receptors.Br J Pharmacol. 2019 May;176(10):1455-1469. doi: 10.1111/bph.14440. Epub 2018 Aug 10. Br J Pharmacol. 2019. PMID: 29981240 Free PMC article.
-
Probing the CB1 Cannabinoid Receptor Binding Pocket with AM6538, a High-Affinity Irreversible Antagonist.Mol Pharmacol. 2019 Nov;96(5):619-628. doi: 10.1124/mol.119.116483. Epub 2019 Sep 12. Mol Pharmacol. 2019. PMID: 31515283 Free PMC article.
-
Use of new approach methodology for hepatic safety assessment of covalent inhibitor drug candidates.Toxicol Res (Camb). 2025 May 25;14(3):tfaf054. doi: 10.1093/toxres/tfaf054. eCollection 2025 Jun. Toxicol Res (Camb). 2025. PMID: 40421426
-
Crystal Structure of the Human Cannabinoid Receptor CB1.Cell. 2016 Oct 20;167(3):750-762.e14. doi: 10.1016/j.cell.2016.10.004. Cell. 2016. PMID: 27768894 Free PMC article.
-
Gαs signalling of the CB1 receptor and the influence of receptor number.Br J Pharmacol. 2017 Aug;174(15):2545-2562. doi: 10.1111/bph.13866. Epub 2017 Jun 19. Br J Pharmacol. 2017. PMID: 28516479 Free PMC article.
References
-
- Di Marzo V. The endocannabinoid system: its general strategy of action, tools for its pharmacological manipulation and potential therapeutic exploration. Pharmacol Res. 2009;60:77–84. - PubMed
-
- Mechoulam R, Parker LA. The endocannabinoid system and the brain. Annu Rev Pscyhol. 2013;64:21–47. - PubMed
-
- Ohno-Shosaku T, Kano M. Endocannabinoid-mediated retrograde modulation of synaptic transmission. Curr Opin Neurobiol. 2014;29:1–8. - PubMed
-
- Ashton CH, Moore PB. Endocannabinoid system dysfunction in mood and related disorders. Acta Psychiatr Scand. 2011;124:250–261. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

