Influenza A Virus NS1 Protein Inhibits the NLRP3 Inflammasome
- PMID: 25978411
- PMCID: PMC4433236
- DOI: 10.1371/journal.pone.0126456
Influenza A Virus NS1 Protein Inhibits the NLRP3 Inflammasome
Erratum in
-
Correction: Influenza A Virus NS1 Protein Inhibits the NLRP3 Inflammasome.PLoS One. 2018 Jul 10;13(7):e0200624. doi: 10.1371/journal.pone.0200624. eCollection 2018. PLoS One. 2018. PMID: 29990347 Free PMC article.
Abstract
The inflammasome is a molecular platform that stimulates the activation of caspase-1 and the processing of pro-interleukin (IL)-1β and pro-IL-18 for secretion. The NOD-like receptor family, pyrin domain containing 3 (NLRP3) protein is activated by diverse molecules and pathogens, leading to the formation of the NLRP3 inflammasome. Recent studies showed that the NLRP3 inflammasome mediates innate immunity against influenza A virus (IAV) infection. In this study, we investigated the function of the IAV non-structural protein 1 (NS1) in the modulation of NLRP3 inflammasome. We found that NS1 proteins derived from both highly pathogenic and low pathogenic strains efficiently decreased secretion of IL-1β and IL-18 from THP-1 cells treated with LPS and ATP. NS1 overexpression significantly impaired the transcription of proinflammatory cytokines by inhibiting transactivation of the nuclear factor-κB (NF-κB), a major transcription activator. Furthermore, NS1 physically interacted with endogenous NLRP3 and activation of the NLRP3 inflammasome was abrogated in NS1-expressing THP-1 cells. These findings suggest that NS1 downregulates NLRP3 inflammasome activation by targeting NLRP3 as well as NF-κB, leading to a reduction in the levels of inflammatory cytokines as a viral immune evasion strategy.
Conflict of interest statement
Figures

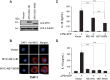


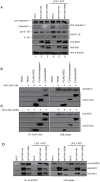
Similar articles
-
The RNA- and TRIM25-Binding Domains of Influenza Virus NS1 Protein Are Essential for Suppression of NLRP3 Inflammasome-Mediated Interleukin-1β Secretion.J Virol. 2016 Mar 28;90(8):4105-4114. doi: 10.1128/JVI.00120-16. Print 2016 Apr. J Virol. 2016. PMID: 26865721 Free PMC article.
-
Aloe vera downregulates LPS-induced inflammatory cytokine production and expression of NLRP3 inflammasome in human macrophages.Mol Immunol. 2013 Dec;56(4):471-9. doi: 10.1016/j.molimm.2013.05.005. Epub 2013 Aug 1. Mol Immunol. 2013. PMID: 23911403
-
NS1 Protein of 2009 Pandemic Influenza A Virus Inhibits Porcine NLRP3 Inflammasome-Mediated Interleukin-1 Beta Production by Suppressing ASC Ubiquitination.J Virol. 2018 Mar 28;92(8):e00022-18. doi: 10.1128/JVI.00022-18. Print 2018 Apr 15. J Virol. 2018. PMID: 29386291 Free PMC article.
-
Interaction between autophagy and the NLRP3 inflammasome.Acta Biochim Biophys Sin (Shanghai). 2019 Nov 18;51(11):1087-1095. doi: 10.1093/abbs/gmz098. Acta Biochim Biophys Sin (Shanghai). 2019. PMID: 31609412 Review.
-
The multifaceted roles of NLRP3-modulating proteins in virus infection.Front Immunol. 2022 Aug 30;13:987453. doi: 10.3389/fimmu.2022.987453. eCollection 2022. Front Immunol. 2022. PMID: 36110852 Free PMC article. Review.
Cited by
-
A systems biology-driven approach to construct a comprehensive protein interaction network of influenza A virus with its host.BMC Infect Dis. 2020 Jul 6;20(1):480. doi: 10.1186/s12879-020-05214-0. BMC Infect Dis. 2020. PMID: 32631335 Free PMC article.
-
The Central Role of Non-Structural Protein 1 (NS1) in Influenza Biology and Infection.Int J Mol Sci. 2020 Feb 22;21(4):1511. doi: 10.3390/ijms21041511. Int J Mol Sci. 2020. PMID: 32098424 Free PMC article. Review.
-
Host Protective Immune Responses against Influenza A Virus Infection.Viruses. 2020 May 3;12(5):504. doi: 10.3390/v12050504. Viruses. 2020. PMID: 32375274 Free PMC article. Review.
-
The interaction between NOLC1 and IAV NS1 protein promotes host cell apoptosis and reduces virus replication.Oncotarget. 2017 Oct 10;8(55):94519-94527. doi: 10.18632/oncotarget.21785. eCollection 2017 Nov 7. Oncotarget. 2017. PMID: 29212246 Free PMC article.
-
Checks and Balances between Autophagy and Inflammasomes during Infection.J Mol Biol. 2018 Jan 19;430(2):174-192. doi: 10.1016/j.jmb.2017.11.006. Epub 2017 Nov 21. J Mol Biol. 2018. PMID: 29162504 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

