Repositioning of Memantine as a Potential Novel Therapeutic Agent against Meningitic E. coli-Induced Pathogenicities through Disease-Associated Alpha7 Cholinergic Pathway and RNA Sequencing-Based Transcriptome Analysis of Host Inflammatory Responses
- PMID: 25993608
- PMCID: PMC4437645
- DOI: 10.1371/journal.pone.0121911
Repositioning of Memantine as a Potential Novel Therapeutic Agent against Meningitic E. coli-Induced Pathogenicities through Disease-Associated Alpha7 Cholinergic Pathway and RNA Sequencing-Based Transcriptome Analysis of Host Inflammatory Responses
Abstract
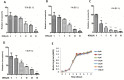
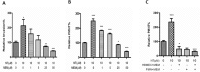
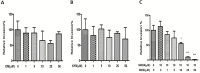
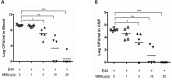
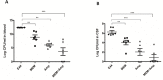
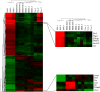
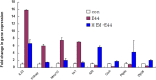
Neonatal sepsis and meningitis (NSM) remains a leading cause worldwide of mortality and morbidity in newborn infants despite the availability of antibiotics over the last several decades. E. coli is the most common gram-negative pathogen causing NSM. Our previous studies show that α7 nicotinic receptor (α7 nAChR), an essential regulator of inflammation, plays a detrimental role in the host defense against NSM. Despite notable successes, there still exists an unmet need for new effective therapeutic approaches to treat this disease. Using the in vitro/in vivo models of the blood-brain barrier (BBB) and RNA-seq, we undertook a drug repositioning study to identify unknown antimicrobial activities for known drugs. We have demonstrated for the first time that memantine (MEM), a FDA-approved drug for treatment of Alzheimer's disease, could very efficiently block E. coli-caused bacteremia and meningitis in a mouse model of NSM in a manner dependent on α7 nAChR. MEM was able to synergistically enhance the antibacterial activity of ampicillin in HBMEC infected with E. coli K1 (E44) and in neonatal mice with E44-caused bacteremia and meningitis. Differential gene expression analysis of RNA-Seq data from mouse BMEC infected with E. coli K1 showed that several E44-increased inflammatory factors, including IL33, IL18rap, MMP10 and Irs1, were significantly reduced by MEM compared to the infected cells without drug treatment. MEM could also significantly up-regulate anti-inflammatory factors, including Tnfaip3, CISH, Ptgds and Zfp36. Most interestingly, these factors may positively and negatively contribute to regulation of NF-κB, which is a hallmark feature of bacterial meningitis. Furthermore, we have demonstrated that circulating BMEC (cBMEC) are the potential novel biomarkers for NSM. MEM could significantly reduce E44-increased blood level of cBMEC in mice. Taken together, our data suggest that memantine can efficiently block host inflammatory responses to bacterial infection through modulation of both inflammatory and anti-inflammatory pathways.
Conflict of interest statement
Figures
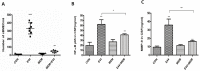

References
-
- Huang SH, Stins M, Kim KS. (2000) Bacterial penetration across the blood-brain barrier during the development of neonatal meningitis. Microbes Infect 2: 1237–1244. - PubMed
-
- Kim KS. (2003) Pathogenesis of bacterial meningitis: from bacteraemia to neuronal injury. Nat Rev Neurosci 4: 376–385. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

