Identification of a Compound That Disrupts Binding of Amyloid-β to the Prion Protein Using a Novel Fluorescence-based Assay
- PMID: 25995455
- PMCID: PMC4505445
- DOI: 10.1074/jbc.M115.637124
Identification of a Compound That Disrupts Binding of Amyloid-β to the Prion Protein Using a Novel Fluorescence-based Assay
Abstract
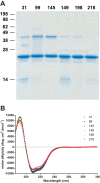
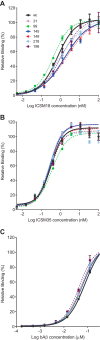
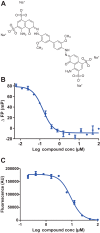
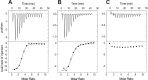
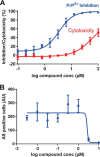
The prion protein (PrP) has been implicated both in prion diseases such as Creutzfeldt-Jakob disease, where its monomeric cellular isoform (PrP(C)) is recruited into pathogenic self-propagating polymers of misfolded protein, and in Alzheimer disease, where PrP(C) may act as a receptor for synaptotoxic oligomeric forms of amyloid-β (Aβ). There has been considerable interest in identification of compounds that bind to PrP(C), stabilizing its native fold and thereby acting as pharmacological chaperones to block prion propagation and pathogenesis. However, compounds binding PrP(C) could also inhibit the binding of toxic Aβ species and may have a role in treating Alzheimer disease, a highly prevalent dementia for which there are currently no disease-modifying treatments. However, the absence of a unitary, readily measurable, physiological function of PrP makes screening for ligands challenging, and the highly heterogeneous nature of Aβ oligomer preparations makes conventional competition binding assays difficult to interpret. We have therefore developed a high-throughput screen that utilizes site-specifically fluorescently labeled protein to identify compounds that bind to PrP and inhibit both Aβ binding and prion propagation. Following a screen of 1,200 approved drugs, we identified Chicago Sky Blue 6B as the first small molecule PrP ligand capable of inhibiting Aβ binding, demonstrating the feasibility of development of drugs to block this interaction. The interaction of Chicago Sky Blue 6B was characterized by isothermal titration calorimetry, and its ability to inhibit Aβ binding and reduce prion levels was established in cell-based assays.
Keywords: Alzheimer disease; amyloid-beta (AB); drug discovery; fluorescence anisotropy; high-throughput screening (HTS); isothermal titration calorimetry (ITC); neurodegenerative disease; prion.
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures


References
-
- Collinge J. (2001) Prion diseases of humans and animals: their causes and molecular basis. Annu. Rev. Neurosci. 24, 519–550 - PubMed
-
- Griffith J. S. (1967) Self replication and scrapie. Nature 215, 1043–1044 - PubMed
-
- Prusiner S. B. (1982) Novel proteinaceous infectious particles cause scrapie. Science 216, 136–144 - PubMed
-
- Sandberg M. K., Al-Doujaily H., Sharps B., Clarke A. R., Collinge J. (2011) Prion propagation and toxicity in vivo occur in two distinct mechanistic phases. Nature 470, 540–542 - PubMed
-
- Sandberg M. K., Al-Doujaily H., Sharps B., De Oliveira M. W., Schmidt C., Richard-Londt A., Lyall S., Linehan J. M., Brandner S., Wadsworth J. D., Clarke A. R., Collinge J. (2014) Prion neuropathology follows the accumulation of alternate prion protein isoforms after infective titre has peaked. Nat. Commun. 5, 4347. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

