The contribution of raised intraneuronal chloride to epileptic network activity
- PMID: 25995461
- PMCID: PMC4438123
- DOI: 10.1523/JNEUROSCI.4105-14.2015
The contribution of raised intraneuronal chloride to epileptic network activity
Abstract
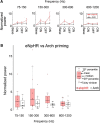
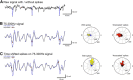
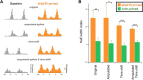
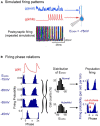
Altered inhibitory function is an important facet of epileptic pathology. A key concept is that GABAergic activity can become excitatory if intraneuronal chloride rises. However, it has proved difficult to separate the role of raised chloride from other contributory factors in complex network phenomena, such as epileptic pathology. Therefore, we asked what patterns of activity are associated with chloride dysregulation by making novel use of Halorhodopsin to load clusters of mouse pyramidal cells artificially with Cl(-). Brief (1-10 s) activation of Halorhodopsin caused substantial positive shifts in the GABAergic reversal potential that were proportional to the charge transfer during the illumination and in adult neocortical pyramidal neurons decayed with a time constant of τ = 8.0 ± 2.8s. At the network level, these positive shifts in EGABA produced a transient rise in network excitability, with many distinctive features of epileptic foci, including high-frequency oscillations with evidence of out-of-phase firing (Ibarz et al., 2010). We show how such firing patterns can arise from quite small shifts in the mean intracellular Cl(-) level, within heterogeneous neuronal populations. Notably, however, chloride loading by itself did not trigger full ictal events, even with additional electrical stimulation to the underlying white matter. In contrast, when performed in combination with low, subepileptic levels of 4-aminopyridine, Halorhodopsin activation rapidly induced full ictal activity. These results suggest that chloride loading has at most an adjunctive role in ictogenesis. Our simulations also show how chloride loading can affect the jitter of action potential timing associated with imminent recruitment to an ictal event (Netoff and Schiff, 2002).
Keywords: chloride; fast spiking interneuron; high frequency oscillations; neocortex; pyramidal neuron; seizure.
Copyright © 2015 Alfonsa et al.
Figures




References
-
- Bernard C, Cossart R, Hirsch JC, Esclapez M, Ben-Ari Y. What is GABAergic inhibition? How is it modified in epilepsy? Epilepsia. 2000;41(Suppl 6):S90–S95. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
