Drosophila Cyclin G and epigenetic maintenance of gene expression during development
- PMID: 25995770
- PMCID: PMC4438588
- DOI: 10.1186/s13072-015-0008-6
Drosophila Cyclin G and epigenetic maintenance of gene expression during development
Abstract
Background: Cyclins and cyclin-dependent kinases (CDKs) are essential for cell cycle regulation and are functionally associated with proteins involved in epigenetic maintenance of transcriptional patterns in various developmental or cellular contexts. Epigenetic maintenance of transcription patterns, notably of Hox genes, requires the conserved Polycomb-group (PcG), Trithorax-group (TrxG), and Enhancer of Trithorax and Polycomb (ETP) proteins, particularly well studied in Drosophila. These proteins form large multimeric complexes that bind chromatin and appose or recognize histone post-translational modifications. PcG genes act as repressors, counteracted by trxG genes that maintain gene activation, while ETPs interact with both, behaving alternatively as repressors or activators. Drosophila Cyclin G negatively regulates cell growth and cell cycle progression, binds and co-localizes with the ETP Corto on chromatin, and participates with Corto in Abdominal-B Hox gene regulation. Here, we address further implications of Cyclin G in epigenetic maintenance of gene expression.
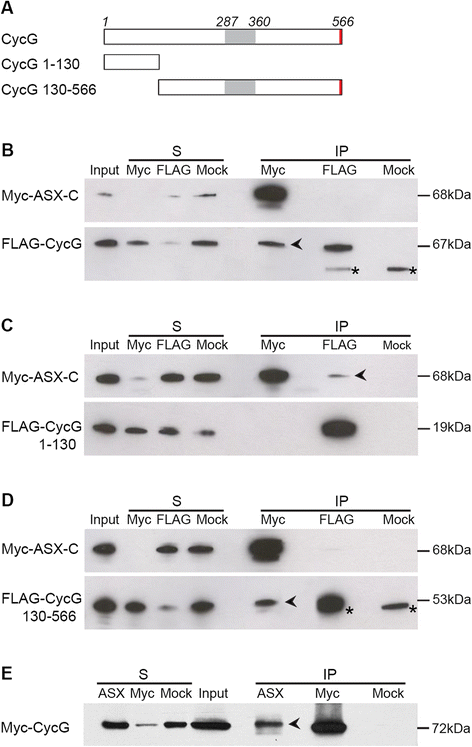
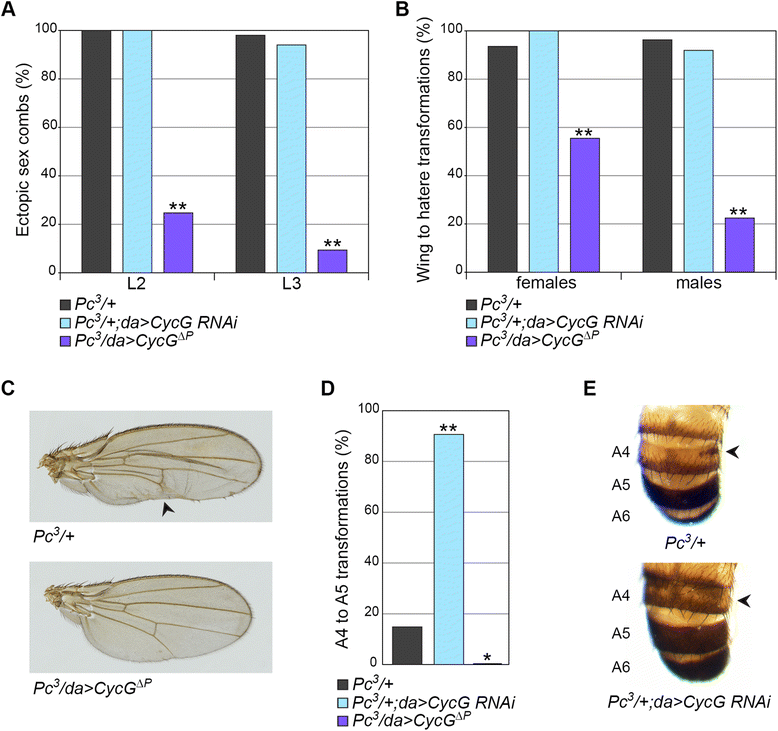
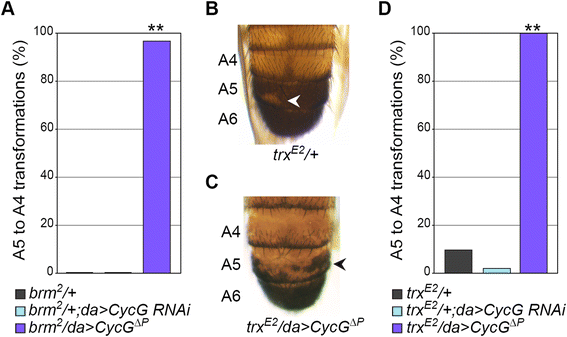
Results: We show that Cyclin G physically interacts and extensively co-localizes on chromatin with the conserved ETP Additional sex combs (ASX), belonging to the repressive PR-DUB complex that participates in H2A deubiquitination and Hox gene silencing. Furthermore, Cyclin G mainly co-localizes with RNA polymerase II phosphorylated on serine 2 that is specific to productive transcription. CycG interacts with Asx, PcG, and trxG genes in Hox gene maintenance, and behaves as a PcG gene. These interactions correlate with modified ectopic Hox protein domains in imaginal discs, consistent with a role for Cyclin G in PcG-mediated Hox gene repression.
Conclusions: We show here that Drosophila CycG is a Polycomb-group gene enhancer, acting in epigenetic maintenance of the Hox genes Sex combs reduced (Scr) and Ultrabithorax (Ubx). However, our data suggest that Cyclin G acts alternatively as a transcriptional activator or repressor depending on the developmental stage, the tissue or the target gene. Interestingly, since Cyclin G interacts with several CDKs, Cyclin G binding to the ETPs ASX or Corto suggests that their activity could depend on Cyclin G-mediated phosphorylation. We discuss whether Cyclin G fine-tunes transcription by controlling H2A ubiquitination and transcriptional elongation via interaction with the ASX subunit of PR-DUB.
Keywords: Cyclin G; Homeotic; Polycomb; Trithorax.
Figures




Similar articles
-
The enhancer of trithorax and polycomb corto interacts with cyclin G in Drosophila.PLoS One. 2008 Feb 20;3(2):e1658. doi: 10.1371/journal.pone.0001658. PLoS One. 2008. PMID: 18286205 Free PMC article.
-
Corto and DSP1 interact and bind to a maintenance element of the Scr Hox gene: understanding the role of Enhancers of trithorax and Polycomb.BMC Biol. 2006 Apr 14;4:9. doi: 10.1186/1741-7007-4-9. BMC Biol. 2006. PMID: 16613610 Free PMC article.
-
Regulation of Abd-B expression by Cyclin G and Corto in the abdominal epithelium of Drosophila.Hereditas. 2008 Jun;145(3):138-46. doi: 10.1111/j.0018-0661.2008.02067.x. Hereditas. 2008. PMID: 18667003
-
Maintenance of Hox gene expression patterns.Adv Exp Med Biol. 2010;689:41-62. doi: 10.1007/978-1-4419-6673-5_3. Adv Exp Med Biol. 2010. PMID: 20795321 Review.
-
[Maintenance of the patterns of expression of homeotic genes in the development of Drosophila melanogaster by proteins of the polycomb, trithorax, and ETP groups].Genetika. 2009 Oct;45(10):1301-18. Genetika. 2009. PMID: 19947541 Review. Russian.
Cited by
-
Ionising radiation exposure-induced regulation of selected biomarkers and their impact in cancer and treatment.Front Nucl Med. 2024 Oct 21;4:1469897. doi: 10.3389/fnume.2024.1469897. eCollection 2024. Front Nucl Med. 2024. PMID: 39498386 Free PMC article. Review.
-
Systemic signalling and local effectors in developmental stability, body symmetry, and size.Cell Stress. 2018 Nov 13;2(12):340-361. doi: 10.15698/cst2018.12.167. Cell Stress. 2018. PMID: 31225459 Free PMC article. Review.
-
Cyclin G and the Polycomb Repressive complexes PRC1 and PR-DUB cooperate for developmental stability.PLoS Genet. 2018 Jul 11;14(7):e1007498. doi: 10.1371/journal.pgen.1007498. eCollection 2018 Jul. PLoS Genet. 2018. PMID: 29995890 Free PMC article.
References
-
- Gopinathan L, Ratnacaram CK, Kaldis P: Established and novel Cdk/cyclin complexes regulating the cell cycle and development. In Results and problems in cell differentiation. Volume 53. Jacek Z. Kubiak; 2011:365–389. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

