Activation of NRG1-ERBB4 signaling potentiates mesenchymal stem cell-mediated myocardial repairs following myocardial infarction
- PMID: 25996292
- PMCID: PMC4669719
- DOI: 10.1038/cddis.2015.91
Activation of NRG1-ERBB4 signaling potentiates mesenchymal stem cell-mediated myocardial repairs following myocardial infarction
Abstract
Mesenchymal stem cell (MSC) transplantation has achieved only modest success in the treatment of ischemic heart disease owing to poor cell viability in the diseased microenvironment. Activation of the NRG1 (neuregulin1)-ERBB4 (v-erb-b2 avian erythroblastic leukemia viral oncogene homolog 4) signaling pathway has been shown to stimulate mature cardiomyocyte cell cycle re-entry and cell division. In this connection, we aimed to determine whether overexpression of ERBB4 in MSCs can enhance their cardio-protective effects following myocardial infarction. NRG1, MSCs or MSC-ERBB4 (MSC with ERBB4 overexpression), were transplanted into mice following myocardial infarction. Superior to that of MSCs and solely NRG1, MSC-ERBB4 transplantation significantly preserved heart functions accompanied with reduced infarct size, enhanced cardiomyocyte division and less apoptosis during early phase of infarction. The transduction of ERBB4 into MSCs indeed increased cell mobility and apoptotic resistance under hypoxic and glucose-deprived conditions via a PI3K/Akt signaling pathway in the presence of NRG1. Unexpectedly, introduction of ERBB4 into MSC in turn potentiates NRG1 synthesis and secretion, thus forming a novel NRG1-ERBB4-NRG1 autocrine loop. Conditioned medium of MSC-ERBB4 containing elevated NRG1, promoted cardiomyocyte growth and division, whereas neutralization of NRG1 blunted this proliferation. These findings collectively suggest that ERBB4 overexpression potentiates MSC survival in the infarcted heart, enhances NRG1 generation to restore declining NRG1 in the infarcted region and stimulates cardiomyocyte division. ERBB4 has an important role in MSC-mediated myocardial repairs.
Figures


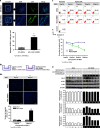

 ), GFP signal, which indicated transfection efficiency, was escalating accordingly, from 1.2% for the first time (d1
), GFP signal, which indicated transfection efficiency, was escalating accordingly, from 1.2% for the first time (d1 ) to 32.7% for the fifth time (d1
) to 32.7% for the fifth time (d1 ). (d2) The expression of NRG1 was upregulated along with increased transfection efficiency. (e) In human 293FT cells, NRG1 expression was upregulated after ERBB4 transduction. The aforementioned experiments were repeated three times and representative images are shown. 293FTe, 293FT expressing pGFP; 293FT-ERBB4, 293FT expressing pER4-GFP
). (d2) The expression of NRG1 was upregulated along with increased transfection efficiency. (e) In human 293FT cells, NRG1 expression was upregulated after ERBB4 transduction. The aforementioned experiments were repeated three times and representative images are shown. 293FTe, 293FT expressing pGFP; 293FT-ERBB4, 293FT expressing pER4-GFP
 ), GFP signal, which indicated transfection efficiency, was escalating accordingly, from 1.2% for the first time (d1
), GFP signal, which indicated transfection efficiency, was escalating accordingly, from 1.2% for the first time (d1 ) to 32.7% for the fifth time (d1
) to 32.7% for the fifth time (d1 ). (d2) The expression of NRG1 was upregulated along with increased transfection efficiency. (e) In human 293FT cells, NRG1 expression was upregulated after ERBB4 transduction. The aforementioned experiments were repeated three times and representative images are shown. 293FTe, 293FT expressing pGFP; 293FT-ERBB4, 293FT expressing pER4-GFP
). (d2) The expression of NRG1 was upregulated along with increased transfection efficiency. (e) In human 293FT cells, NRG1 expression was upregulated after ERBB4 transduction. The aforementioned experiments were repeated three times and representative images are shown. 293FTe, 293FT expressing pGFP; 293FT-ERBB4, 293FT expressing pER4-GFP

References
-
- Tse H F, Kwong YL, Chan JK, Lo G, Ho CL, Lau CP et al. Angiogenesis in ischaemic myocardium by intramyocardial autologous bone marrow mononuclear cell implantation. Lancet 2003; 361: 47–49. - PubMed
-
- Psaltis P J, Spoon D B, Wong D T. Utility of mesenchymal stromal cells for myocardial infarction. Transitioning from bench to bedside. Minerva Cardioangiol 2013; 61: 639–663. - PubMed
-
- Zhang Y, Liang X, Lian Q, Tse H F. Perspective and challenges of mesenchymal stem cells for cardiovascular regeneration. Exp Rev Cardiovasc Ther 2013; 11: 505–517. - PubMed
-
- Lopes JP, Fiarresga A, Silva PC, Feliciano J, Cruz RF. Mesenchymal stem cell therapy in heart disease. Rev Port Cardiol 2013; 32: 43–47. - PubMed
-
- Chang W, Song BW, Lim S, Song H, Shim CY, Cha MJ et al. Mesenchymal stem cells pretreated with delivered Hph-1-Hsp70 protein are protected from hypoxia-mediated cell death and rescue heart functions from myocardial injury. Stem Cell 2009; 27: 2283–2292. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

