14-3-3 isoforms bind directly exon B of the 5'-UTR of human surfactant protein A2 mRNA
- PMID: 26001776
- PMCID: PMC4504974
- DOI: 10.1152/ajplung.00088.2015
14-3-3 isoforms bind directly exon B of the 5'-UTR of human surfactant protein A2 mRNA
Abstract
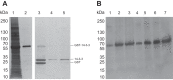
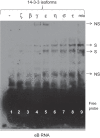
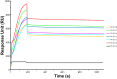
Human surfactant protein (SP) A (SP-A), an innate immunity molecule, is encoded by two genes, SFTPA1 and SFTPA2. The 5'-untranslated splice variant of SP-A2 (ABD), but not SP-A1 (AD), contains exon B (eB). eB is an enhancer for transcription and translation and contains cis-regulatory elements. Specific trans-acting factors, including 14-3-3, bind eB. The 14-3-3 protein family contains seven isoforms that have been found by mass spectrometry in eB electromobility shift assays (Noutsios et al. Am J Physiol Lung Cell Mol Physiol 304: L722-L735, 2013). We used four different approaches to investigate whether 14-3-3 isoforms bind directly to eB. 1) eB RNA pulldown assays showed that 14-3-3 isoforms specifically bind eB. 2) RNA electromobility shift assay complexes were formed using purified 14-3-3 isoforms β, γ, ε, η, σ, and τ, but not isoform ζ, with wild-type eB RNA. 3 and 4) RNA affinity chromatography assays and surface plasmon resonance analysis showed that 14-3-3 isoforms β, γ, ε, η, σ, and τ, but not isoform ζ, specifically and directly bind eB. Inhibition of 14-3-3 isoforms γ, ε, η, and τ/θ with shRNAs in NCI-H441 cells resulted in downregulation of SP-A2 levels but did not affect SP-A1 levels. However, inhibition of 14-3-3 isoform σ was correlated with lower levels of SP-A1 and SP-A2. Inhibition of 14-3-3 isoform ζ/δ, which does not bind eB, had no effect on expression levels of SP-A1 and SP-A2. In conclusion, the 14-3-3 protein family affects differential regulation of SP-A1 and SP-A2 by binding directly to SP-A2 5'-UTR mRNA.
Keywords: 14-3-3; 5′-untranslated region; human pulmonary surfactant protein A; translation.
Copyright © 2015 the American Physiological Society.
Figures



References
-
- Aitken A, Howell S, Jones D, Madrazo J, Patel Y. 14-3-3 α and δ are the phosphorylated forms of raf-activating 14-3-3 β and ζ. In vivo stoichiometric phosphorylation in brain at a Ser-Pro-Glu-Lys MOTIF. J Biol Chem 270: 5706–5709, 1995. - PubMed
-
- Asdaghi N, Kilani RT, Hosseini-Tabatabaei A, Odemuyiwa SO, Hackett TL, Knight DA, Ghahary A, Moqbel R. Extracellular 14-3-3 from human lung epithelial cells enhances MMP-1 expression. Mol Cell Biochem 360: 261–270, 2012. - PubMed
-
- Bortner JD, Das A, Umstead TM, Freeman WM, Somiari R, Aliaga C, Phelps DS, El-Bayoumy K. Down-regulation of 14-3-3 isoforms and annexin A5 proteins in lung adenocarcinoma induced by the tobacco-specific nitrosamine NNK in the A/J mouse revealed by proteomic analysis. J Proteome Res 8: 4050–4061, 2009. - PMC - PubMed
-
- Bridges D, Moorhead GB. 14-3-3 proteins: a number of functions for a numbered protein. Sci STKE 2004: re10, 2004. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

