Structure Guided Chemical Modifications of Propylthiouracil Reveal Novel Small Molecule Inhibitors of Cytochrome b5 Reductase 3 That Increase Nitric Oxide Bioavailability
- PMID: 26001785
- PMCID: PMC4505432
- DOI: 10.1074/jbc.M114.629964
Structure Guided Chemical Modifications of Propylthiouracil Reveal Novel Small Molecule Inhibitors of Cytochrome b5 Reductase 3 That Increase Nitric Oxide Bioavailability
Abstract
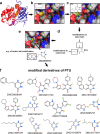
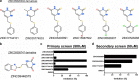
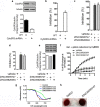
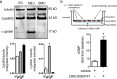
NADH cytochrome b5 reductase 3 (CYB5R3) is critical for reductive reactions such as fatty acid elongation, cholesterol biosynthesis, drug metabolism, and methemoglobin reduction. Although the physiological and metabolic importance of CYB5R3 has been established in hepatocytes and erythrocytes, emerging investigations suggest that CYB5R3 is critical for nitric oxide signaling and vascular function. However, advancement toward fully understanding CYB5R3 function has been limited due to a lack of potent small molecule inhibitors. Because of this restriction, we modeled the binding mode of propylthiouracil, a weak inhibitor of CYB5R3 (IC50 = ∼275 μM), and used it as a guide to predict thiouracil-biased inhibitors from the set of commercially available compounds in the ZINC database. Using this approach, we validated two new potent derivatives of propylthiouracil, ZINC05626394 (IC50 = 10.81 μM) and ZINC39395747 (IC50 = 9.14 μM), both of which inhibit CYB5R3 activity in cultured cells. Moreover, we found that ZINC39395747 significantly increased NO bioavailability in renal vascular cells, augmented renal blood flow, and decreased systemic blood pressure in response to vasoconstrictors in spontaneously hypertensive rats. These compounds will serve as a new tool to examine the biological functions of CYB5R3 in physiology and disease and also as a platform for new drug development.
Keywords: computational biology; cytochrome b5 reductase 3; hemoglobin; nitric oxide; oxidation-reduction (redox); vascular.
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures

Similar articles
-
Loss of smooth muscle CYB5R3 amplifies angiotensin II-induced hypertension by increasing sGC heme oxidation.JCI Insight. 2019 Oct 3;4(19):e129183. doi: 10.1172/jci.insight.129183. JCI Insight. 2019. PMID: 31487266 Free PMC article.
-
External mitochondrial NADH-dependent reductase of redox cyclers: VDAC1 or Cyb5R3?Free Radic Biol Med. 2014 Sep;74:74-84. doi: 10.1016/j.freeradbiomed.2014.06.005. Epub 2014 Jun 16. Free Radic Biol Med. 2014. PMID: 24945955
-
Familial Congenital Methemoglobinemia in Pomeranian Dogs Caused by a Missense Variant in the NADH-Cytochrome B5 Reductase Gene.J Vet Intern Med. 2018 Jan;32(1):165-171. doi: 10.1111/jvim.15031. Epub 2018 Jan 22. J Vet Intern Med. 2018. PMID: 29356095 Free PMC article.
-
Cytochrome b5 reductases: Redox regulators of cell homeostasis.J Biol Chem. 2022 Dec;298(12):102654. doi: 10.1016/j.jbc.2022.102654. Epub 2022 Oct 29. J Biol Chem. 2022. PMID: 36441026 Free PMC article. Review.
-
Cellular and Molecular Mechanisms of Recessive Hereditary Methaemoglobinaemia Type II.J Clin Med. 2018 Oct 10;7(10):341. doi: 10.3390/jcm7100341. J Clin Med. 2018. PMID: 30309019 Free PMC article. Review.
Cited by
-
Escorting α-globin to eNOS: α-globin-stabilizing protein paves the way.J Clin Invest. 2018 Nov 1;128(11):4755-4757. doi: 10.1172/JCI124302. Epub 2018 Oct 8. J Clin Invest. 2018. PMID: 30295642 Free PMC article.
-
Identification of metabolism-related subtypes and feature genes in Alzheimer's disease.J Transl Med. 2023 Sep 15;21(1):628. doi: 10.1186/s12967-023-04324-y. J Transl Med. 2023. PMID: 37715200 Free PMC article.
-
The role of globins in cardiovascular physiology.Physiol Rev. 2022 Apr 1;102(2):859-892. doi: 10.1152/physrev.00037.2020. Epub 2021 Sep 6. Physiol Rev. 2022. PMID: 34486392 Free PMC article. Review.
-
Data, Reagents, Assays and Merits of Proteomics for SARS-CoV-2 Research and Testing.Mol Cell Proteomics. 2020 Sep;19(9):1503-1522. doi: 10.1074/mcp.RA120.002164. Epub 2020 Jun 26. Mol Cell Proteomics. 2020. PMID: 32591346 Free PMC article.
-
Ordered Motions in the Nitric-Oxide Dioxygenase Mechanism of Flavohemoglobin and Assorted Globins with Tightly Coupled Reductases.Adv Exp Med Biol. 2023;1414:45-96. doi: 10.1007/5584_2022_751. Adv Exp Med Biol. 2023. PMID: 36520413
References
-
- Reddy V. V., Kupfer D., Caspi E. (1977) Mechanism of C-5 double bond introduction in the biosynthesis of cholesterol by rat liver microsomes. J. Biol. Chem. 252, 2797–2801 - PubMed
-
- Hildebrandt A., Estabrook R. W. (1971) Evidence for the participation of cytochrome b 5 in hepatic microsomal mixed-function oxidation reactions. Arch. Biochem. Biophys. 143, 66–79 - PubMed
-
- Passon P. G., Hultquist D. E. (1972) Soluble cytochrome b5 reductase from human erythrocytes. Biochim. Biophys. Acta 275, 62–73 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

