Marine Macrocyclic Imines, Pinnatoxins A and G: Structural Determinants and Functional Properties to Distinguish Neuronal α7 from Muscle α1(2)βγδ nAChRs
- PMID: 26004441
- PMCID: PMC4461042
- DOI: 10.1016/j.str.2015.04.009
Marine Macrocyclic Imines, Pinnatoxins A and G: Structural Determinants and Functional Properties to Distinguish Neuronal α7 from Muscle α1(2)βγδ nAChRs
Abstract
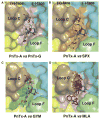
Pinnatoxins are macrocyclic imine phycotoxins associated with algal blooms and shellfish toxicity. Functional analysis of pinnatoxin A and pinnatoxin G by binding and voltage-clamp electrophysiology on membrane-embedded neuronal α7, α4β2, α3β2, and muscle-type α12βγδ nicotinic acetylcholine receptors (nAChRs) reveals high-affinity binding and potent antagonism for the α7 and α12βγδ subtypes. The toxins also bind to the nAChR surrogate, acetylcholine-binding protein (AChBP), with low Kd values reflecting slow dissociation. Crystal structures of pinnatoxin-AChBP complexes (1.9-2.2 Å resolution) show the multiple anchoring points of the hydrophobic portion, the cyclic imine, and the substituted bis-spiroketal and cyclohexene ring systems of the pinnatoxins that dictate tight binding between the opposing loops C and F at the receptor subunit interface, as observed for the 13-desmethyl-spirolide C and gymnodimine A congeners. Uniquely, however, the bulky bridged EF-ketal ring specific to the pinnatoxins extends radially from the interfacial-binding pocket to interact with the sequence-variable loop F and govern nAChR subtype selectivity and central neurotoxicity.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures
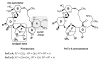


Comment in
-
From shellfish poisoning to neuroscience.Structure. 2015 Jun 2;23(6):979-80. doi: 10.1016/j.str.2015.05.008. Structure. 2015. PMID: 26039347
References
-
- Brejc K, van Dijk WJ, Klaassen RV, Schuurmans M, van Der Oost J, Smit AB, Sixma TK. Crystal structure of an ACh-binding protein reveals the ligand-binding domain of nicotinic receptors. Nature. 2001;411:269–276. - PubMed
-
- CCP4. The CCP4 suite: Programs for protein crystallography. Acta Crystallogr D Biol Crystallogr. 1994;50:760–763. - PubMed
-
- Celie PH, van Rossum-Fikkert SE, van Dijk WJ, Brejc K, Smit AB, Sixma TK. Nicotine and carbamylcholine binding to nicotinic acetylcholine receptors as studied in AChBP crystal structures. Neuron. 2004;41:907–914. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

