Slitrk5 Mediates BDNF-Dependent TrkB Receptor Trafficking and Signaling
- PMID: 26004511
- PMCID: PMC4784688
- DOI: 10.1016/j.devcel.2015.04.009
Slitrk5 Mediates BDNF-Dependent TrkB Receptor Trafficking and Signaling
Abstract
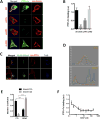
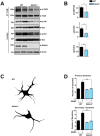
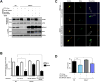
Recent studies in humans and in genetic mouse models have identified Slit- and NTRK-like family (Slitrks) as candidate genes for neuropsychiatric disorders. All Slitrk isotypes are highly expressed in the CNS, where they mediate neurite outgrowth, synaptogenesis, and neuronal survival. However, the molecular mechanisms underlying these functions are not known. Here, we report that Slitrk5 modulates brain-derived neurotrophic factor (BDNF)-dependent biological responses through direct interaction with TrkB receptors. Under basal conditions, Slitrk5 interacts primarily with a transsynaptic binding partner, protein tyrosine phosphatase δ (PTPδ); however, upon BDNF stimulation, Slitrk5 shifts to cis-interactions with TrkB. In the absence of Slitrk5, TrkB has a reduced rate of ligand-dependent recycling and altered responsiveness to BDNF treatment. Structured illumination microscopy revealed that Slitrk5 mediates optimal targeting of TrkB receptors to Rab11-positive recycling endosomes through recruitment of a Rab11 effector protein, Rab11-FIP3. Thus, Slitrk5 acts as a TrkB co-receptor that mediates its BDNF-dependent trafficking and signaling.
Copyright © 2015 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no competing financial interests.
Figures




Similar articles
-
Retrolinkin cooperates with endophilin A1 to mediate BDNF-TrkB early endocytic trafficking and signaling from early endosomes.Mol Biol Cell. 2011 Oct;22(19):3684-98. doi: 10.1091/mbc.E11-04-0308. Epub 2011 Aug 17. Mol Biol Cell. 2011. PMID: 21849472 Free PMC article.
-
Myosin Va mediates BDNF-induced postendocytic recycling of full-length TrkB and its translocation into dendritic spines.J Cell Sci. 2015 Mar 15;128(6):1108-22. doi: 10.1242/jcs.160259. Epub 2015 Jan 27. J Cell Sci. 2015. PMID: 25632160
-
BDNF-dependent recycling facilitates TrkB translocation to postsynaptic density during LTP via a Rab11-dependent pathway.J Neurosci. 2013 May 22;33(21):9214-30. doi: 10.1523/JNEUROSCI.3256-12.2013. J Neurosci. 2013. PMID: 23699532 Free PMC article.
-
The Rab11-regulated endocytic pathway and BDNF/TrkB signaling: Roles in plasticity changes and neurodegenerative diseases.Neurobiol Dis. 2022 Sep;171:105796. doi: 10.1016/j.nbd.2022.105796. Epub 2022 Jun 18. Neurobiol Dis. 2022. PMID: 35728773 Review.
-
Regulation of TrkB cell surface expression-a mechanism for modulation of neuronal responsiveness to brain-derived neurotrophic factor.Cell Tissue Res. 2020 Oct;382(1):5-14. doi: 10.1007/s00441-020-03224-7. Epub 2020 Jun 15. Cell Tissue Res. 2020. PMID: 32556728 Free PMC article. Review.
Cited by
-
Slitrk1 is localized to excitatory synapses and promotes their development.Sci Rep. 2016 Jun 7;6:27343. doi: 10.1038/srep27343. Sci Rep. 2016. PMID: 27273464 Free PMC article.
-
Coordination of AMPA receptor trafficking by Rab GTPases.Small GTPases. 2019 Nov;10(6):419-432. doi: 10.1080/21541248.2017.1337546. Epub 2017 Jul 5. Small GTPases. 2019. PMID: 28628388 Free PMC article. Review.
-
Speech- and language-linked FOXP2 mutation targets protein motors in striatal neurons.Brain. 2023 Aug 1;146(8):3542-3557. doi: 10.1093/brain/awad090. Brain. 2023. PMID: 37137515 Free PMC article.
-
Neurotrophin signaling endosomes: biogenesis, regulation, and functions.Curr Opin Neurobiol. 2016 Aug;39:139-45. doi: 10.1016/j.conb.2016.06.004. Epub 2016 Jun 18. Curr Opin Neurobiol. 2016. PMID: 27327126 Free PMC article. Review.
-
Genome-wide DNA methylation profiling of primary colorectal laterally spreading tumors identifies disease-specific epimutations on common pathways.Int J Cancer. 2018 Nov 15;143(10):2488-2498. doi: 10.1002/ijc.31765. Epub 2018 Sep 19. Int J Cancer. 2018. PMID: 30183087 Free PMC article.
References
-
- Abelson JF, Kwan KY, O’Roak BJ, Baek DY, Stillman AA, Morgan TM, Mathews CA, Pauls DL, Rasin MR, Gunel M, et al. Sequence variants in SLITRK1 are associated with Tourette’s syndrome. Science. 2005;310:317–320. - PubMed
-
- Arevalo JC, Waite J, Rajagopal R, Beyna M, Chen ZY, Lee FS, Chao MV. Cell survival through Trk neurotrophin receptors is differentially regulated by ubiquitination. Neuron. 2006;50:549–559. - PubMed
-
- Aruga J, Mikoshiba K. Identification and characterization of Slitrk, a novel neuronal transmembrane protein family controlling neurite outgrowth. Molecular and cellular neurosciences. 2003;24:117–129. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- MH079513/MH/NIMH NIH HHS/United States
- R01 GM034107/GM/NIGMS NIH HHS/United States
- 5UL1TR000457/TR/NCATS NIH HHS/United States
- P50 MH079513/MH/NIMH NIH HHS/United States
- RC1 AI080309/AI/NIAID NIH HHS/United States
- R01 NS052819/NS/NINDS NIH HHS/United States
- R01 EY008538/EY/NEI NIH HHS/United States
- GM804302/GM/NIGMS NIH HHS/United States
- R01 HL097797/HL/NHLBI NIH HHS/United States
- R01 NS030687/NS/NINDS NIH HHS/United States
- AI080309/AI/NIAID NIH HHS/United States
- HL097797/HL/NHLBI NIH HHS/United States
- NS052819/NS/NINDS NIH HHS/United States
- HL66592/HL/NHLBI NIH HHS/United States
- NS030687/NS/NINDS NIH HHS/United States
- UL1 TR000457/TR/NCATS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

