Vitamin D3-dependent VDR signaling delays ron-mediated breast tumorigenesis through suppression of β-catenin activity
- PMID: 26008979
- PMCID: PMC4599271
- DOI: 10.18632/oncotarget.4059
Vitamin D3-dependent VDR signaling delays ron-mediated breast tumorigenesis through suppression of β-catenin activity
Abstract
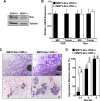
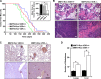
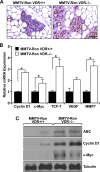
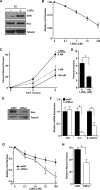
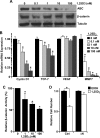
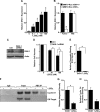
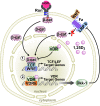
The Ron receptor is upregulated in human breast cancers and correlates with enhanced metastasis and reduced patient survival. Ron overexpression drives mammary tumorigenesis through direct β-catenin activation and augmented tumor cell proliferation, migration and invasion. Ron and β-catenin are also coordinately elevated in breast cancers. The vitamin D receptor (VDR) antagonizes β-catenin signaling. Herein, we examined mammary tumor onset and progression using a Ron-driven murine model of breast tumorigenesis crossed with VDR deficient mice. VDR ablation accelerated mammary tumor onset and led to tumors that exhibited a desmoplastic phenotype and enhanced metastases. Tumor levels of active β-catenin were markedly increased in the absence of VDR. In vitro, VDR activation in breast cancer cells reduced β-catenin activation and transcriptional activity leading to elevated expression of the extracellular Wnt inhibitor dickkopf-related protein 1, and a reduction in the interaction of β-catenin with the cyclin D1 promoter. Expression of a stabilized form or β-catenin ablated the protective effects of VDR activation.Collectively, these studies delineate a protective role for VDR signaling in Ron-induced mammary tumorigenesis through disruption of β-catenin activation.
Keywords: breast cancer; ron; vitamin D3 receptor; β-catenin.
Conflict of interest statement
There is no conflict of interest.
Figures
Similar articles
-
Conditional deletion of β-catenin in mammary epithelial cells of Ron receptor, Mst1r, overexpressing mice alters mammary tumorigenesis.Endocrinology. 2012 Jun;153(6):2735-46. doi: 10.1210/en.2011-1543. Epub 2012 Apr 2. Endocrinology. 2012. PMID: 22474186 Free PMC article.
-
β-Catenin is required for Ron receptor-induced mammary tumorigenesis.Oncogene. 2011 Aug 25;30(34):3694-704. doi: 10.1038/onc.2011.86. Epub 2011 Mar 21. Oncogene. 2011. PMID: 21423209 Free PMC article.
-
Vitamin D receptor prevents tumour development by regulating the Wnt/β-catenin signalling pathway in human colorectal cancer.BMC Cancer. 2023 Apr 12;23(1):336. doi: 10.1186/s12885-023-10690-z. BMC Cancer. 2023. PMID: 37046222 Free PMC article.
-
Vitamin D and Wnt/beta-catenin pathway in colon cancer: role and regulation of DICKKOPF genes.Anticancer Res. 2008 Sep-Oct;28(5A):2613-23. Anticancer Res. 2008. PMID: 19035286 Review.
-
1α,25(OH)2-dihydroxyvitamin D3/VDR protects the skin from UVB-induced tumor formation by interacting with the β-catenin pathway.J Steroid Biochem Mol Biol. 2013 Jul;136:229-32. doi: 10.1016/j.jsbmb.2012.09.024. Epub 2012 Sep 28. J Steroid Biochem Mol Biol. 2013. PMID: 23026511 Free PMC article. Review.
Cited by
-
Wnt and Vitamin D at the Crossroads in Solid Cancer.Cancers (Basel). 2020 Nov 19;12(11):3434. doi: 10.3390/cancers12113434. Cancers (Basel). 2020. PMID: 33227961 Free PMC article. Review.
-
Impact of Western and Mediterranean Diets and Vitamin D on Muscle Fibers of Sedentary Rats.Nutrients. 2018 Feb 17;10(2):231. doi: 10.3390/nu10020231. Nutrients. 2018. PMID: 29462978 Free PMC article.
-
Emerging perspectives: unraveling the anticancer potential of vitamin D3.Naunyn Schmiedebergs Arch Pharmacol. 2024 May;397(5):2877-2933. doi: 10.1007/s00210-023-02819-5. Epub 2023 Nov 23. Naunyn Schmiedebergs Arch Pharmacol. 2024. PMID: 37994947 Review.
-
The regulatory role of miRNAs on VDR in breast cancer.Transcription. 2017 Aug 8;8(4):232-241. doi: 10.1080/21541264.2017.1317695. Epub 2017 Jun 9. Transcription. 2017. PMID: 28598255 Free PMC article. Review.
-
Loss of the vitamin D receptor in human breast and prostate cancers strongly induces cell apoptosis through downregulation of Wnt/β-catenin signaling.Bone Res. 2017 Sep 5;5:17023. doi: 10.1038/boneres.2017.23. eCollection 2017. Bone Res. 2017. PMID: 28944088 Free PMC article.
References
-
- Maggiora P, Marchio S, Stella MC, Giai M, Belfiore A, De Bortoli M, Di Renzo MF, Costantino A, Sismondi P, Comoglio PM. Overexpression of the RON gene in human breast carcinoma. Oncogene. 1998;16:2927–2933. - PubMed
-
- Lee WY, Chen HH, Chow NH, Su WC, Lin PW, Guo HR. Prognostic significance of co-expression of RON and MET receptors in node-negative breast cancer patients. Clin Cancer Res. 2005;11:2222–2228. - PubMed
-
- Welm AL, Sneddon JB, Taylor C, Nuyten DS, van de Vijver MJ, Hasegawa BH, Bishop JM. The macrophage-stimulating protein pathway promotes metastasis in a mouse model for breast cancer and predicts poor prognosis in humans. Proceedings of the National Academy of Sciences of the United States of America. 2007;104:7570–7575. - PMC - PubMed
-
- Kretschmann KL, Eyob H, Buys SS, Welm AL. The macrophage stimulating protein/Ron pathway as a potential therapeutic target to impede multiple mechanisms involved in breast cancer progression. Current drug targets. 2010;11:1157–1168. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

