Ryanodine receptor gating controls generation of diastolic calcium waves in cardiac myocytes
- PMID: 26009544
- PMCID: PMC4442793
- DOI: 10.1085/jgp.201411281
Ryanodine receptor gating controls generation of diastolic calcium waves in cardiac myocytes
Abstract
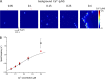
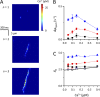
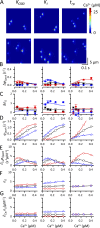
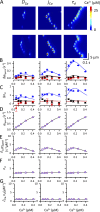
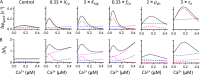
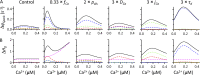
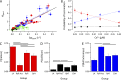
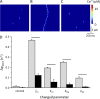
The role of cardiac ryanodine receptor (RyR) gating in the initiation and propagation of calcium waves was investigated using a mathematical model comprising a stochastic description of RyR gating and a deterministic description of calcium diffusion and sequestration. We used a one-dimensional array of equidistantly spaced RyR clusters, representing the confocal scanning line, to simulate the formation of calcium sparks. Our model provided an excellent description of the calcium dependence of the frequency of diastolic calcium sparks and of the increased tendency for the production of calcium waves after a decrease in cytosolic calcium buffering. We developed a hypothesis relating changes in the propensity to form calcium waves to changes of RyR gating and tested it by simulation. With a realistic RyR gating model, increased ability of RyR to be activated by Ca2+ strongly increased the propensity for generation of calcium waves at low (0.05-0.1-µM) calcium concentrations but only slightly at high (0.2-0.4-µM) calcium concentrations. Changes in RyR gating altered calcium wave formation by changing the calcium sensitivity of spontaneous calcium spark activation and/or the average number of open RyRs in spontaneous calcium sparks. Gating changes that did not affect RyR activation by Ca2+ had only a weak effect on the propensity to form calcium waves, even if they strongly increased calcium spark frequency. Calcium waves induced by modulating the properties of the RyR activation site could be suppressed by inhibiting the spontaneous opening of the RyR. These data can explain the increased tendency for production of calcium waves under conditions when RyR gating is altered in cardiac diseases.
© 2015 Petrovič et al.
Figures

Similar articles
-
Frequency and release flux of calcium sparks in rat cardiac myocytes: a relation to RYR gating.J Gen Physiol. 2010 Jul;136(1):101-16. doi: 10.1085/jgp.200910380. Epub 2010 Jun 14. J Gen Physiol. 2010. PMID: 20548054 Free PMC article.
-
Size Matters: Ryanodine Receptor Cluster Size Affects Arrhythmogenic Sarcoplasmic Reticulum Calcium Release.J Am Heart Assoc. 2018 Jun 21;7(13):e008724. doi: 10.1161/JAHA.118.008724. J Am Heart Assoc. 2018. PMID: 29929992 Free PMC article.
-
Role of ryanodine receptor cooperativity in Ca2+-wave-mediated triggered activity in cardiomyocytes.J Physiol. 2024 Dec;602(24):6745-6787. doi: 10.1113/JP286145. Epub 2024 Nov 20. J Physiol. 2024. PMID: 39565684
-
Ca2+ stores regulate ryanodine receptor Ca2+ release channels via luminal and cytosolic Ca2+ sites.Clin Exp Pharmacol Physiol. 2007 Sep;34(9):889-96. doi: 10.1111/j.1440-1681.2007.04708.x. Clin Exp Pharmacol Physiol. 2007. PMID: 17645636 Review.
-
Putting out the fire: what terminates calcium-induced calcium release in cardiac muscle?Cell Calcium. 2004 Jun;35(6):591-601. doi: 10.1016/j.ceca.2004.01.013. Cell Calcium. 2004. PMID: 15110149 Review.
Cited by
-
The Interplay of Rogue and Clustered Ryanodine Receptors Regulates Ca2+ Waves in Cardiac Myocytes.Front Physiol. 2018 Apr 26;9:393. doi: 10.3389/fphys.2018.00393. eCollection 2018. Front Physiol. 2018. PMID: 29755362 Free PMC article.
-
Of bipolar cell synapses, light-activated K+ channels, and substrate binding to DAT.J Gen Physiol. 2015 Jul;146(1):1-2. doi: 10.1085/jgp.201511449. J Gen Physiol. 2015. PMID: 26123193 Free PMC article. No abstract available.
-
A Stochastic Spatiotemporal Model of Rat Ventricular Myocyte Calcium Dynamics Demonstrated Necessary Features for Calcium Wave Propagation.Membranes (Basel). 2021 Dec 18;11(12):989. doi: 10.3390/membranes11120989. Membranes (Basel). 2021. PMID: 34940490 Free PMC article.
-
Ca2+ dynamics in interstitial cells: foundational mechanisms for the motor patterns in the gastrointestinal tract.Physiol Rev. 2024 Jan 1;104(1):329-398. doi: 10.1152/physrev.00036.2022. Epub 2023 Aug 10. Physiol Rev. 2024. PMID: 37561138 Free PMC article. Review.
-
Cardiac calcium dysregulation in mice with chronic kidney disease.J Cell Mol Med. 2020 Mar;24(6):3669-3677. doi: 10.1111/jcmm.15066. Epub 2020 Feb 16. J Cell Mol Med. 2020. PMID: 32064746 Free PMC article.
References
-
- Bai Y., Jones P.P., Guo J., Zhong X., Clark R.B., Zhou Q., Wang R., Vallmitjana A., Benitez R., Hove-Madsen L., et al. . 2013. Phospholamban knockout breaks arrhythmogenic Ca2+ waves and suppresses catecholaminergic polymorphic ventricular tachycardia in mice. Circ. Res. 113:517–526. 10.1161/CIRCRESAHA.113.301678 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

