Serotonin release from the neuronal cell body and its long-lasting effects on the nervous system
- PMID: 26009775
- PMCID: PMC4455765
- DOI: 10.1098/rstb.2014.0196
Serotonin release from the neuronal cell body and its long-lasting effects on the nervous system
Abstract
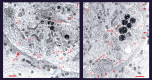
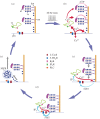
Serotonin, a modulator of multiple functions in the nervous system, is released predominantly extrasynaptically from neuronal cell bodies, axons and dendrites. This paper describes how serotonin is released from cell bodies of Retzius neurons in the central nervous system (CNS) of the leech, and how it affects neighbouring glia and neurons. The large Retzius neurons contain serotonin packed in electrodense vesicles. Electrical stimulation with 10 impulses at 1 Hz fails to evoke exocytosis from the cell body, but the same number of impulses at 20 Hz promotes exocytosis via a multistep process. Calcium entry into the neuron triggers calcium-induced calcium release, which activates the transport of vesicle clusters to the plasma membrane. Exocytosis occurs there for several minutes. Serotonin that has been released activates autoreceptors that induce an inositol trisphosphate-dependent calcium increase, which produces further exocytosis. This positive feedback loop subsides when the last vesicles in the cluster fuse and calcium returns to basal levels. Serotonin released from the cell body is taken up by glia and released elsewhere in the CNS. Synchronous bursts of neuronal electrical activity appear minutes later and continue for hours. In this way, a brief train of impulses is translated into a long-term modulation in the nervous system.
Keywords: extrasynaptic; neuron–glia communication; serotonergic modulation; serotonin; somatic exocytosis; transmitter release.
Figures

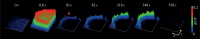

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
