Immunolocalization of G protein-coupled estrogen receptor in the rat epididymis
- PMID: 26012959
- PMCID: PMC4450610
- DOI: 10.1186/s12958-015-0042-z
Immunolocalization of G protein-coupled estrogen receptor in the rat epididymis
Abstract
Background: Estrogen plays an important role in male reproduction, and males lacking estrogen signaling in the reproductive tissues are infertile. Estrogen signaling is mediated via two nuclear receptors, ERα and ERβ, but it was recently found that a G protein-coupled estrogen receptor (GPER) is present in the testis. It is believed that GPER is a membrane form of the estrogen receptor and mediates non-classical estrogen signaling. However, the cellular localization of GPER in the epididymis is unknown. Therefore, the objective of this study was to determine the cellular and regional expression of GPER in the rat epididymis.
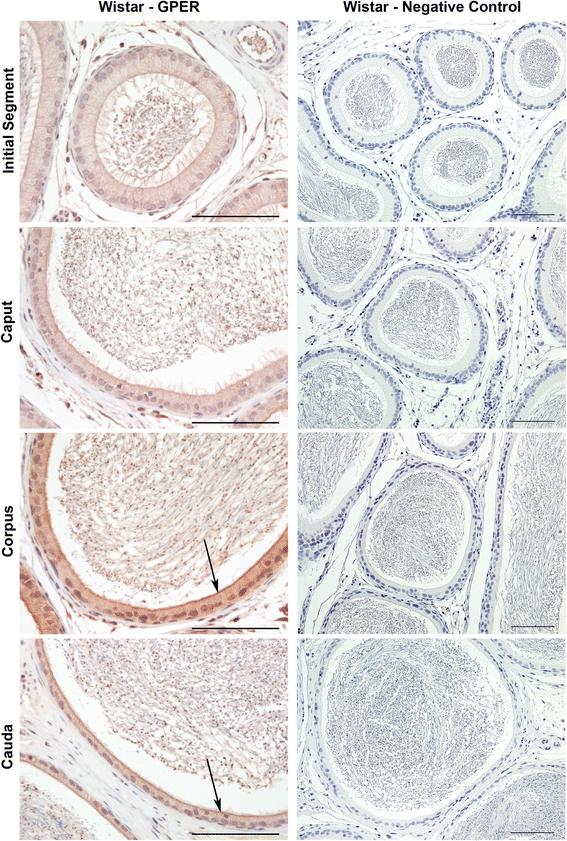
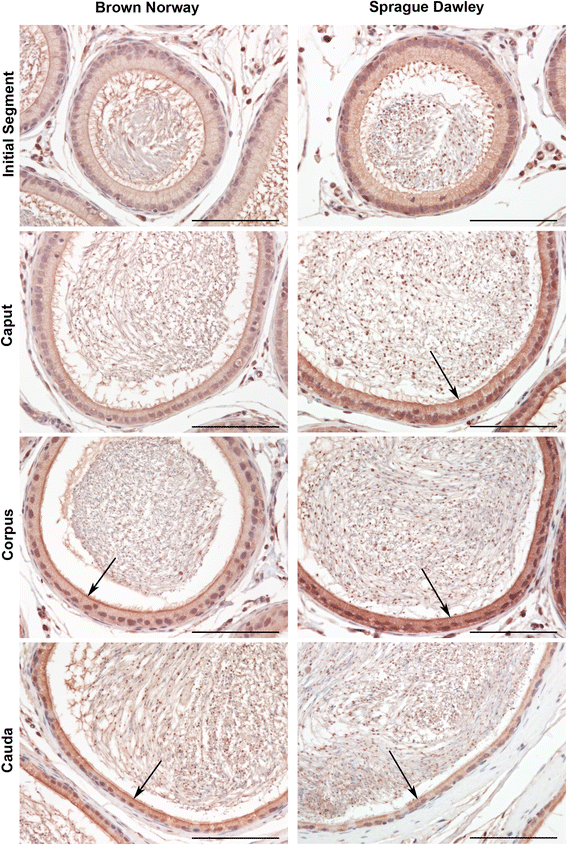
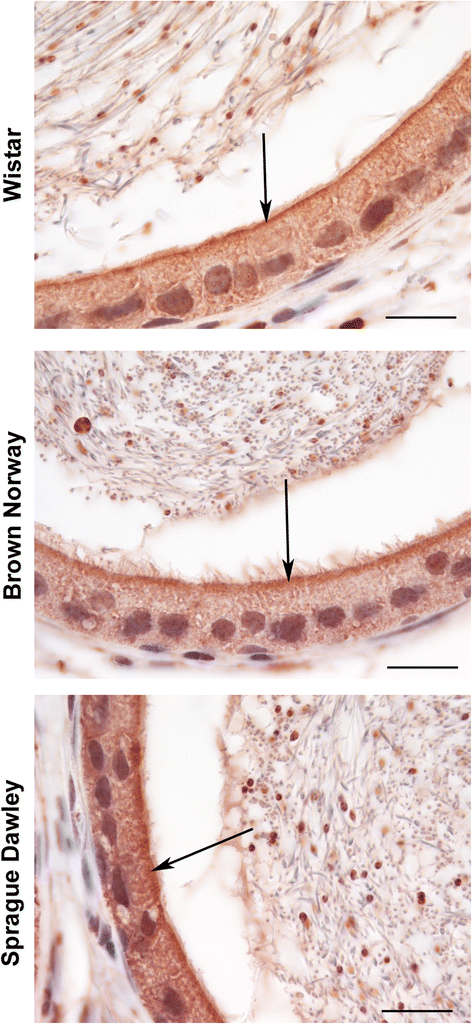
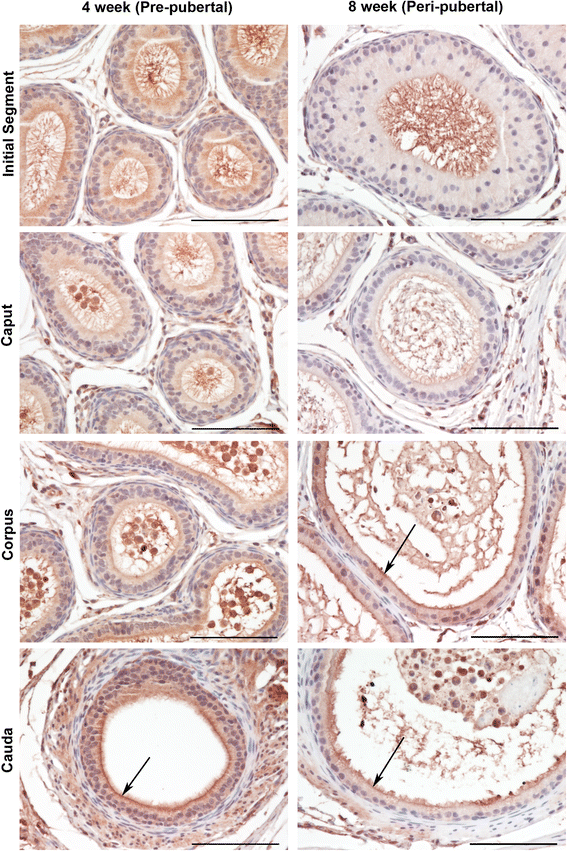
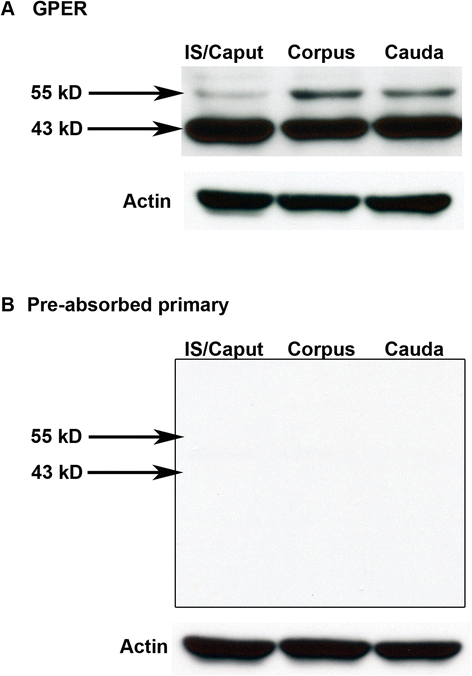
Findings: To localize expression, immunohistochemistry (IHC) was performed using fixed epididymal tissue. Three strains and ages of rats were used to identify whether GPER expression is strain or age specific. Our results are the first to demonstrate immunostaining of GPER in epididymal epithelial cells. Expression was highest near the apical membrane followed by the cytoplasm, consistent with a membrane bound receptor. The highest expression in adult rats was observed in corpus followed by cauda. Western blotting analysis of epididymal tissues from Sprague Dawley rats confirmed specificity of the antibody and regional expression.
Conclusions: Expression of GPER in the corpus and cauda suggests a role for non-classical estrogen signaling in sperm maturation in the corpus, and sperm protection/storage in the cauda. GPER expression pre-pubertally suggests that estrogen may have a role in epithelial cell development in addition to regulation of adult function.
Figures
Similar articles
-
Age-, cell- and region-specific immunoexpression of estrogen receptor alpha (but not estrogen receptor beta) during postnatal development of the epididymis and vas deferens of the rat and disruption of this pattern by neonatal treatment with diethylstilbestrol.Endocrinology. 2001 Feb;142(2):874-86. doi: 10.1210/endo.142.2.7978. Endocrinology. 2001. PMID: 11159861
-
Estrogen and its receptors in efferent ductules and epididymis.J Androl. 2011 Nov-Dec;32(6):600-13. doi: 10.2164/jandrol.110.012872. Epub 2011 Mar 25. J Androl. 2011. PMID: 21441425 Review.
-
G protein-coupled estrogen receptor (GPER) in adult boar testes, epididymis and spermatozoa during epididymal maturation.Int J Biol Macromol. 2018 Sep;116:113-119. doi: 10.1016/j.ijbiomac.2018.05.015. Epub 2018 May 4. Int J Biol Macromol. 2018. PMID: 29730010
-
Immunolocalization of G Protein-Coupled Estrogen Receptor in the Pig Epididymis.Anat Rec (Hoboken). 2018 Aug;301(8):1467-1473. doi: 10.1002/ar.23837. Epub 2018 May 7. Anat Rec (Hoboken). 2018. PMID: 29679442
-
Twenty years of the G protein-coupled estrogen receptor GPER: Historical and personal perspectives.J Steroid Biochem Mol Biol. 2018 Feb;176:4-15. doi: 10.1016/j.jsbmb.2017.03.021. Epub 2017 Mar 25. J Steroid Biochem Mol Biol. 2018. PMID: 28347854 Free PMC article. Review.
Cited by
-
Role of GPER-Mediated Signaling in Testicular Functions and Tumorigenesis.Cells. 2020 Sep 17;9(9):2115. doi: 10.3390/cells9092115. Cells. 2020. PMID: 32957524 Free PMC article. Review.
-
Missing Information from the Estrogen Receptor Puzzle: Where Are They Localized in Bull Reproductive Tissues and Spermatozoa?Cells. 2020 Jan 10;9(1):183. doi: 10.3390/cells9010183. Cells. 2020. PMID: 31936899 Free PMC article.
-
Phthalate Exposure During Pregnancy and Lactation Transgenerationally Impairs the Epididymis in the Offspring of Rats.J Biochem Mol Toxicol. 2025 Jul;39(7):e70379. doi: 10.1002/jbt.70379. J Biochem Mol Toxicol. 2025. PMID: 40551524 Free PMC article.
-
Correlation of functional and radioligand binding characteristics of GPER ligands confirming aldosterone as a GPER agonist.Pharmacol Res Perspect. 2022 Oct;10(5):e00995. doi: 10.1002/prp2.995. Pharmacol Res Perspect. 2022. PMID: 36065843 Free PMC article.
-
Expression of G-Protein-Coupled Estrogen Receptor (GPER) in Whole Testicular Tissue and Laser-Capture Microdissected Testicular Compartments of Men with Normal and Aberrant Spermatogenesis.Biology (Basel). 2022 Feb 26;11(3):373. doi: 10.3390/biology11030373. Biology (Basel). 2022. PMID: 35336747 Free PMC article.
References
-
- Robaire B, Hinton B, Orgebin-Crist M-C. The Epididymis. In: Neill JD, editor. Knobil and Neill's Physiology of Reproduction. Volume 1. Third. New York: Elsevier; 2006. pp. 1071–1147.
-
- Cooper TG. Epididymis and sperm function. Andrologia. 1996;28(Suppl 1):57–59. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

