The hormonal peptide Elabela guides angioblasts to the midline during vasculogenesis
- PMID: 26017639
- PMCID: PMC4468421
- DOI: 10.7554/eLife.06726
The hormonal peptide Elabela guides angioblasts to the midline during vasculogenesis
Abstract
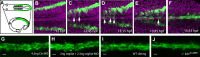
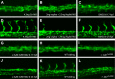
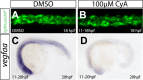
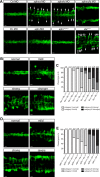
A key step in the de novo formation of the embryonic vasculature is the migration of endothelial precursors, the angioblasts, to the position of the future vessels. To form the first axial vessels, angioblasts migrate towards the midline and coalesce underneath the notochord. Vascular endothelial growth factor has been proposed to serve as a chemoattractant for the angioblasts and to regulate this medial migration. Here we challenge this model and instead demonstrate that angioblasts rely on their intrinsic expression of Apelin receptors (Aplr, APJ) for their migration to the midline. We further show that during this angioblast migration Apelin receptor signaling is mainly triggered by the recently discovered ligand Elabela (Ela). As neither of the ligands Ela or Apelin (Apln) nor their receptors have previously been implicated in regulating angioblast migration, we hereby provide a novel mechanism for regulating vasculogenesis, with direct relevance to physiological and pathological angiogenesis.
Keywords: Vegf; angioblast migration; chemoattractant; developmental biology; dorsal aorta; endothelial cell migration; human biology; medicine; notochord; stem cells; zebrafish.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures


Similar articles
-
Somite morphogenesis is required for axial blood vessel formation during zebrafish embryogenesis.Elife. 2022 Feb 9;11:e74821. doi: 10.7554/eLife.74821. Elife. 2022. PMID: 35137687 Free PMC article.
-
Endothelial cell-specific chemotaxis receptor (ecscr) promotes angioblast migration during vasculogenesis and enhances VEGF receptor sensitivity.Blood. 2010 Jun 3;115(22):4614-22. doi: 10.1182/blood-2009-10-248856. Epub 2010 Jan 19. Blood. 2010. PMID: 20086248 Free PMC article.
-
The LIM-homeodomain transcription factor Islet2a promotes angioblast migration.Dev Biol. 2016 Jun 15;414(2):181-92. doi: 10.1016/j.ydbio.2016.04.019. Epub 2016 Apr 25. Dev Biol. 2016. PMID: 27126199
-
Endothelial precursor cell migration during vasculogenesis.Circ Res. 2007 Jul 20;101(2):125-36. doi: 10.1161/CIRCRESAHA.107.148932. Circ Res. 2007. PMID: 17641236 Review.
-
The role of FGF and VEGF in angioblast induction and migration during vascular development.Dev Dyn. 2001 Jan;220(1):1-17. doi: 10.1002/1097-0177(2000)9999:9999<::AID-DVDY1087>3.0.CO;2-2. Dev Dyn. 2001. PMID: 11146503 Review.
Cited by
-
The apelinergic system: a perspective on challenges and opportunities in cardiovascular and metabolic disorders.Ann N Y Acad Sci. 2019 Nov;1455(1):12-33. doi: 10.1111/nyas.14123. Epub 2019 Jun 25. Ann N Y Acad Sci. 2019. PMID: 31236974 Free PMC article. Review.
-
Svep1 is a binding ligand of Tie1 and affects specific aspects of facial lymphatic development in a Vegfc-independent manner.Elife. 2023 Apr 25;12:e82969. doi: 10.7554/eLife.82969. Elife. 2023. PMID: 37097004 Free PMC article.
-
MicroRNA 139-5p coordinates APLNR-CXCR4 crosstalk during vascular maturation.Nat Commun. 2016 Apr 12;7:11268. doi: 10.1038/ncomms11268. Nat Commun. 2016. PMID: 27068353 Free PMC article.
-
Expression and functional implications of the renal apelinergic system in rodents.PLoS One. 2017 Aug 17;12(8):e0183094. doi: 10.1371/journal.pone.0183094. eCollection 2017. PLoS One. 2017. PMID: 28817612 Free PMC article.
-
Regulatory pathways governing murine coronary vessel formation are dysregulated in the injured adult heart.Nat Commun. 2019 Jul 22;10(1):3276. doi: 10.1038/s41467-019-10710-2. Nat Commun. 2019. PMID: 31332177 Free PMC article.
References
-
- Charo DN, Ho M, Fajardo G, Kawana M, Kundu RK, Sheikh AY, Finsterbach TP, Leeper NJ, Ernst KV, Chen MM, Ho YD, Chun HJ, Bernstein D, Ashley EA, Quertermous T. Endogenous regulation of cardiovascular function by apelin-APJ. American Journal of Physiology. Heart and Circulatory Physiology. 2009;297:H1904–H1913. doi: 10.1152/ajpheart.00686.2009. - DOI - PMC - PubMed
-
- Cleaver O, Krieg PA. VEGF mediates angioblast migration during development of the dorsal aorta in Xenopus. Development. 1998;125:3905–3914. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

