Progesterone treatment in two rat models of ocular ischemia
- PMID: 26024074
- PMCID: PMC4419778
- DOI: 10.1167/iovs.14-16070
Progesterone treatment in two rat models of ocular ischemia
Abstract
Purpose: To determine whether the neurosteroid progesterone, shown to have protective effects in animal models of traumatic brain injury, stroke, and spinal cord injury, is also protective in ocular ischemia animal models.
Methods: Progesterone treatment was tested in two ocular ischemia models in rats: a rodent anterior ischemic optic neuropathy (rAION) model, which induces permanent monocular optic nerve stroke, and the middle cerebral artery occlusion (MCAO) model, which causes transient ischemia in both the retina and brain due to an intraluminal filament that blocks the ophthalmic and middle cerebral arteries. Visual function and retinal histology were assessed to determine whether progesterone attenuated retinal injury in these models. Additionally, behavioral testing and 2% 2,3,5-triphenyltetrazolium chloride (TTC) staining in brains were used to compare progesterone's neuroprotective effects in both retina and brain using the MCAO model.
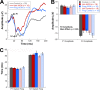
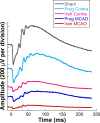
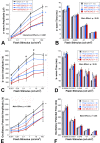
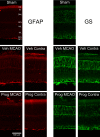
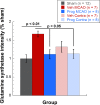
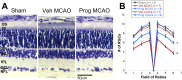
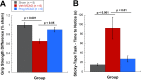
Results: Progesterone treatment showed no effect on visual evoked potential (VEP) reduction and retinal ganglion cell loss in the permanent rAION model. In the transient MCAO model, progesterone treatment reduced (1) electroretinogram (ERG) deficits, (2) MCAO-induced upregulation of glutamine synthetase (GS) and glial fibrillary acidic protein (GFAP), and (3) retinal ganglion cell loss. As expected, progesterone treatment also had significant protective effects in behavioral tests and a reduction in infarct size in the brain.
Conclusions: Progesterone treatment showed protective effects in the retina following MCAO but not rAION injury, which may result from mechanistic differences with injury type and the therapeutic action of progesterone.
Figures



Similar articles
-
Progesterone treatment shows greater protection in brain vs. retina in a rat model of middle cerebral artery occlusion: Progesterone receptor levels may play an important role.Restor Neurol Neurosci. 2016 Nov 22;34(6):947-963. doi: 10.3233/RNN-160672. Restor Neurol Neurosci. 2016. PMID: 27802245 Free PMC article.
-
Neuroprotective effects of recombinant human granulocyte colony-stimulating factor (G-CSF) in a rat model of anterior ischemic optic neuropathy (rAION).Exp Eye Res. 2014 Jan;118:109-16. doi: 10.1016/j.exer.2013.11.012. Epub 2013 Dec 5. Exp Eye Res. 2014. PMID: 24316388
-
Severity of middle cerebral artery occlusion determines retinal deficits in rats.Exp Neurol. 2014 Apr;254:206-15. doi: 10.1016/j.expneurol.2014.02.005. Epub 2014 Feb 8. Exp Neurol. 2014. PMID: 24518488 Free PMC article.
-
[Nonarteritic ischemic optic neuropathy animal model and its treatment applications].Nippon Ganka Gakkai Zasshi. 2014 Apr;118(4):331-61. Nippon Ganka Gakkai Zasshi. 2014. PMID: 24864434 Review. Japanese.
-
Retinal and optic nerve ischemia.Continuum (Minneap Minn). 2014 Aug;20(4 Neuro-ophthalmology):838-56. doi: 10.1212/01.CON.0000453315.82884.a1. Continuum (Minneap Minn). 2014. PMID: 25099097 Free PMC article. Review.
Cited by
-
Ischemic Optic Neuropathy: A Review of Current and Potential Future Pharmacotherapies.Pharmaceuticals (Basel). 2024 Sep 27;17(10):1281. doi: 10.3390/ph17101281. Pharmaceuticals (Basel). 2024. PMID: 39458922 Free PMC article. Review.
-
Inherited Retinal Dystrophies: Role of Oxidative Stress and Inflammation in Their Physiopathology and Therapeutic Implications.Antioxidants (Basel). 2022 May 30;11(6):1086. doi: 10.3390/antiox11061086. Antioxidants (Basel). 2022. PMID: 35739983 Free PMC article. Review.
-
Long-Term Functional and Structural Consequences of Primary Blast Overpressure to the Eye.J Neurotrauma. 2018 Sep 1;35(17):2104-2116. doi: 10.1089/neu.2017.5394. Epub 2018 Jul 2. J Neurotrauma. 2018. PMID: 29648979 Free PMC article.
-
Brain-derived neuerotrophic factor and related mechanisms that mediate and influence progesterone-induced neuroprotection.Front Endocrinol (Lausanne). 2024 Feb 26;15:1286066. doi: 10.3389/fendo.2024.1286066. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 38469139 Free PMC article. Review.
-
Contribution of GABAa, GABAc and glycine receptors to rat dark-adapted oscillatory potentials in the time and frequency domain.Oncotarget. 2017 Sep 8;8(44):77696-77709. doi: 10.18632/oncotarget.20770. eCollection 2017 Sep 29. Oncotarget. 2017. PMID: 29100418 Free PMC article.
References
-
- Benavente O,, Eliasziw M, Streifler JY,, et al. Prognosis after transient monocular blindness associated with carotid-artery stenosis. N Engl J Med. 2001; 345: 1084–1090. - PubMed
-
- Mead GE, Lewis SC,, Wardlaw JM, Dennis MS.Comparison of risk factors in patients with transient and prolonged eye and brain ischemic syndromes. Stroke. 2002; 33: 2383–2390. - PubMed
-
- Slepyan DH, Rankin RM,, Stahler C, Jr, Gibbons GE.Amaurosis fugax: a clinical comparison. Stroke. 1975; 6: 493–496. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

