Identification of 53 compounds that block Ebola virus-like particle entry via a repurposing screen of approved drugs
- PMID: 26038505
- PMCID: PMC4317638
- DOI: 10.1038/emi.2014.88
Identification of 53 compounds that block Ebola virus-like particle entry via a repurposing screen of approved drugs
Abstract
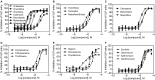
In light of the current outbreak of Ebola virus disease, there is an urgent need to develop effective therapeutics to treat Ebola infection, and drug repurposing screening is a potentially rapid approach for identifying such therapeutics. We developed a biosafety level 2 (BSL-2) 1536-well plate assay to screen for entry inhibitors of Ebola virus-like particles (VLPs) containing the glycoprotein (GP) and the matrix VP40 protein fused to a beta-lactamase reporter protein and applied this assay for a rapid drug repurposing screen of Food and Drug Administration (FDA)-approved drugs. We report here the identification of 53 drugs with activity of blocking Ebola VLP entry into cells. These 53 active compounds can be divided into categories including microtubule inhibitors, estrogen receptor modulators, antihistamines, antipsychotics, pump/channel antagonists, and anticancer/antibiotics. Several of these compounds, including microtubule inhibitors and estrogen receptor modulators, had previously been reported to be active in BSL-4 infectious Ebola virus replication assays and in animal model studies. Our assay represents a robust, effective and rapid high-throughput screen for the identification of lead compounds in drug development for the treatment of Ebola virus infection.
Keywords: Antipsychotics; Ebola virus; Ebola virus glycoprotein; VP40; drug repurposing screen; estrogen receptor modulator; microtubule inhibitor; virus entry.
Figures

References
-
- Cohen J. Infectious disease. Ebola vaccine: little and late. Science. 2014;345:1441–1442. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
