Microglia disrupt mesolimbic reward circuitry in chronic pain
- PMID: 26041913
- PMCID: PMC4452552
- DOI: 10.1523/JNEUROSCI.4036-14.2015
Microglia disrupt mesolimbic reward circuitry in chronic pain
Abstract
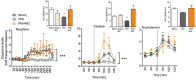
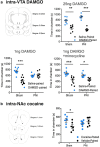
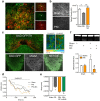
Chronic pain attenuates midbrain dopamine (DA) transmission, as evidenced by a decrease in opioid-evoked DA release in the ventral striatum, suggesting that the occurrence of chronic pain impairs reward-related behaviors. However, mechanisms by which pain modifies DA transmission remain elusive. Using in vivo microdialysis and microinjection of drugs into the mesolimbic DA system, we demonstrate in mice and rats that microglial activation in the VTA compromises not only opioid-evoked release of DA, but also other DA-stimulating drugs, such as cocaine. Our data show that loss of stimulated extracellular DA is due to impaired chloride homeostasis in midbrain GABAergic interneurons. Treatment with minocycline or interfering with BDNF signaling restored chloride transport within these neurons and recovered DA-dependent reward behavior. Our findings demonstrate that a peripheral nerve injury causes activated microglia within reward circuitry that result in disruption of dopaminergic signaling and reward behavior. These results have broad implications that are not restricted to the problem of pain, but are also relevant to affective disorders associated with disruption of reward circuitry. Because chronic pain causes glial activation in areas of the CNS important for mood and affect, our findings may translate to other disorders, including anxiety and depression, that demonstrate high comorbidity with chronic pain.
Keywords: addiction; affective disorder; chronic pain; dopamine; emotion; opioids.
Copyright © 2015 Taylor et al.
Figures


References
-
- Brettschneider J, Toledo JB, Van Deerlin VM, Elman L, McCluskey L, Lee VM, Trojanowski JQ. Microglial activation correlates with disease progression and upper motor neuron clinical symptoms in amyotrophic lateral sclerosis. PLoS One. 2012;7:e39216. doi: 10.1371/journal.pone.0039216. - DOI - PMC - PubMed
