Activin A inhibits BMP-signaling by binding ACVR2A and ACVR2B
- PMID: 26047946
- PMCID: PMC4467681
- DOI: 10.1186/s12964-015-0104-z
Activin A inhibits BMP-signaling by binding ACVR2A and ACVR2B
Abstract
Background: Activins are members of the TGF-β family of ligands that have multiple biological functions in embryonic stem cells as well as in differentiated tissue. Serum levels of activin A were found to be elevated in pathological conditions such as cachexia, osteoporosis and cancer. Signaling by activin A through canonical ALK4-ACVR2 receptor complexes activates the transcription factors SMAD2 and SMAD3. Activin A has a strong affinity to type 2 receptors, a feature that they share with some of the bone morphogenetic proteins (BMPs). Activin A is also elevated in myeloma patients with advanced disease and is involved in myeloma bone disease.
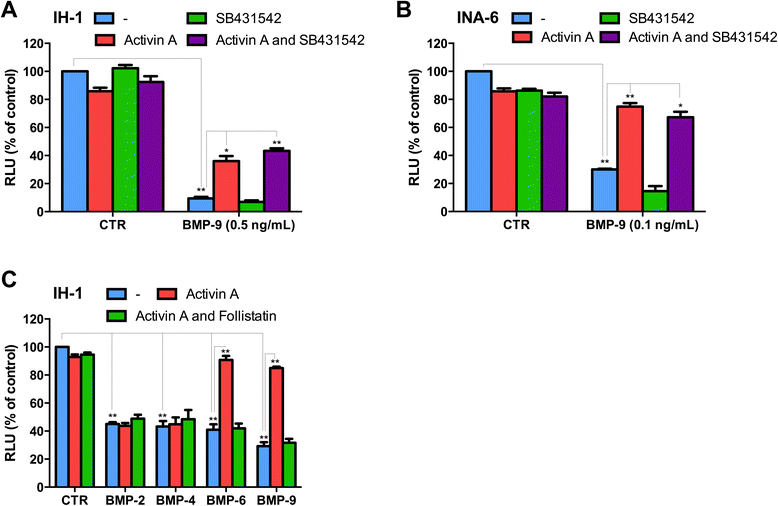
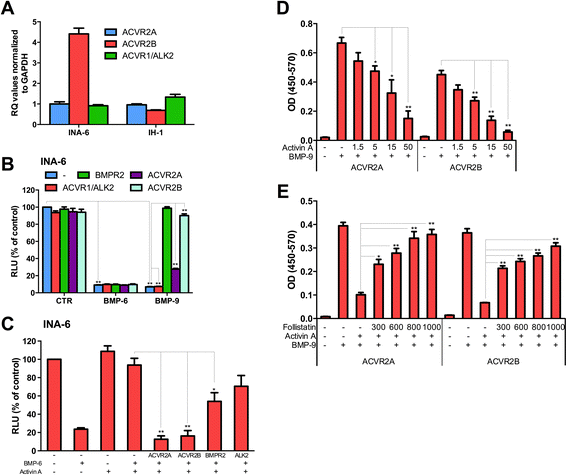
Results: In this study we investigated effects of activin A binding to receptors that are shared with BMPs using myeloma cell lines with well-characterized BMP-receptor expression and responses. Activin A antagonized BMP-6 and BMP-9, but not BMP-2 and BMP-4. Activin A was able to counteract BMPs that signal through the type 2 receptors ACVR2A and ACVR2B in combination with ALK2, but not BMPs that signal through BMPR2 in combination with ALK3 and ALK6.
Conclusions: We propose that one important way that activin A regulates cell behavior is by antagonizing BMP-ACVR2A/ACVR2B/ALK2 signaling.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

