The Treatment of Cushing's Disease
- PMID: 26067718
- PMCID: PMC4523083
- DOI: 10.1210/er.2013-1048
The Treatment of Cushing's Disease
Abstract
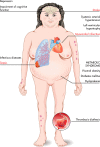
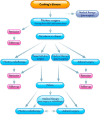
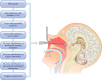
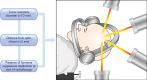
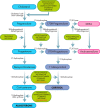
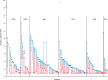
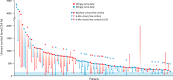
Cushing's disease (CD), or pituitary-dependent Cushing's syndrome, is a severe endocrine disease caused by a corticotroph pituitary tumor and associated with increased morbidity and mortality. The first-line treatment for CD is pituitary surgery, which is followed by disease remission in around 78% and relapse in around 13% of patients during the 10-year period after surgery, so that nearly one third of patients experience in the long-term a failure of surgery and require an additional second-line treatment. Patients with persistent or recurrent CD require additional treatments, including pituitary radiotherapy, adrenal surgery, and/or medical therapy. Pituitary radiotherapy is effective in controlling cortisol excess in a large percentage of patients, but it is associated with a considerable risk of hypopituitarism. Adrenal surgery is followed by a rapid and definitive control of cortisol excess in nearly all patients, but it induces adrenal insufficiency. Medical therapy has recently acquired a more important role compared to the past, due to the recent employment of novel compounds able to control cortisol secretion or action. Currently, medical therapy is used as a presurgical treatment, particularly for severe disease; or as postsurgical treatment, in cases of failure or incomplete surgical tumor resection; or as bridging therapy before, during, and after radiotherapy while waiting for disease control; or, in selected cases, as primary therapy, mainly when surgery is not an option. The adrenal-directed drug ketoconazole is the most commonly used drug, mainly because of its rapid action, whereas the glucocorticoid receptor antagonist, mifepristone, is highly effective in controlling clinical comorbidities, mainly glucose intolerance, thus being a useful treatment for CD when it is associated with diabetes mellitus. Pituitary-directed drugs have the advantage of acting at the site responsible for CD, the pituitary tumor. Among this group of drugs, the dopamine agonist cabergoline and the somatostatin analog pasireotide result in disease remission in a consistent subgroup of patients with CD. Recently, pasireotide has been approved for the treatment of CD when surgery has failed or when surgery is not an option, and mifepristone has been approved for the treatment of Cushing's syndrome when associated with impairment of glucose metabolism in case of the lack of a surgical indication. Recent experience suggests that the combination of different drugs may be able to control cortisol excess in a great majority of patients with CD.
Figures

References
-
- Pivonello R, De Martino MC, De Leo M, Lombardi G, Colao A. Cushing's syndrome. Endocrinol Metab Clin North Am. 2008;37(1):135–149; ix. - PubMed
-
- Newell-Price J, Bertagna X, Grossman AB, Nieman LK. Cushing's syndrome. Lancet. 2006;367(9522):1605–1617. - PubMed
-
- Steffensen C, Bak AM, Rubeck KZ, Jørgensen JO. Epidemiology of Cushing's syndrome. Neuroendocrinology. 2010;92(suppl 1):1–5. - PubMed
-
- Arnaldi G, Angeli A, Atkinson AB, et al. Diagnosis and complications of Cushing's syndrome: a consensus statement. J Clin Endocrinol Metab. 2003;88(12):5593–5602. - PubMed
-
- Newell-Price J, Trainer P, Besser M, Grossman A. The diagnosis and differential diagnosis of Cushing's syndrome and pseudo-Cushing's states. Endocr Rev. 1998;19(5):647–672. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

