Combined Targeted DNA Sequencing in Non-Small Cell Lung Cancer (NSCLC) Using UNCseq and NGScopy, and RNA Sequencing Using UNCqeR for the Detection of Genetic Aberrations in NSCLC
- PMID: 26076459
- PMCID: PMC4468211
- DOI: 10.1371/journal.pone.0129280
Combined Targeted DNA Sequencing in Non-Small Cell Lung Cancer (NSCLC) Using UNCseq and NGScopy, and RNA Sequencing Using UNCqeR for the Detection of Genetic Aberrations in NSCLC
Abstract
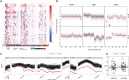
The recent FDA approval of the MiSeqDx platform provides a unique opportunity to develop targeted next generation sequencing (NGS) panels for human disease, including cancer. We have developed a scalable, targeted panel-based assay termed UNCseq, which involves a NGS panel of over 200 cancer-associated genes and a standardized downstream bioinformatics pipeline for detection of single nucleotide variations (SNV) as well as small insertions and deletions (indel). In addition, we developed a novel algorithm, NGScopy, designed for samples with sparse sequencing coverage to detect large-scale copy number variations (CNV), similar to human SNP Array 6.0 as well as small-scale intragenic CNV. Overall, we applied this assay to 100 snap-frozen lung cancer specimens lacking same-patient germline DNA (07-0120 tissue cohort) and validated our results against Sanger sequencing, SNP Array, and our recently published integrated DNA-seq/RNA-seq assay, UNCqeR, where RNA-seq of same-patient tumor specimens confirmed SNV detected by DNA-seq, if RNA-seq coverage depth was adequate. In addition, we applied the UNCseq assay on an independent lung cancer tumor tissue collection with available same-patient germline DNA (11-1115 tissue cohort) and confirmed mutations using assays performed in a CLIA-certified laboratory. We conclude that UNCseq can identify SNV, indel, and CNV in tumor specimens lacking germline DNA in a cost-efficient fashion.
Conflict of interest statement
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

