The hypoxic tumor microenvironment: A driving force for breast cancer progression
- PMID: 26079100
- PMCID: PMC4678039
- DOI: 10.1016/j.bbamcr.2015.05.036
The hypoxic tumor microenvironment: A driving force for breast cancer progression
Abstract
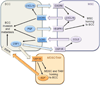
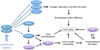
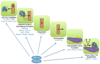
Intratumoral hypoxia is a common finding in breast cancer and is associated with a significantly increased risk of metastasis and patient mortality. Hypoxia-inducible factors activate the transcription of a large battery of genes encoding proteins that promote primary tumor vascularization and growth, stromal cell recruitment, extracellular matrix remodeling, premetastatic niche formation, cell motility, local tissue invasion, extravasation at sites of metastasis, and maintenance of the cancer stem cell phenotype that is required to generate secondary tumors. Recent preclinical studies suggest that the combination of cytotoxic chemotherapy with drugs that inhibit hypoxia-inducible factors may improve outcome for women with triple-negative breast cancer. This article is part of a Special Issue entitled: Tumor Microenvironment Regulation of Cancer Cell Survival, Metastasis, Inflammation, and Immune Surveillance edited by Peter Ruvolo and Gregg L. Semenza.
Keywords: Bone metastasis; Lung metastasis; Lymph node metastasis; Mesenchymal stem cells; Microvesicles; Myeloid-derived suppressor cells; Tumor-associated macrophages.
Copyright © 2015 Elsevier B.V. All rights reserved.
Figures


References
-
- Hanahan D, Coussens LM. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell. 2012;21:309–322. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

