Microglia Activate Migration of Glioma Cells through a Pyk2 Intracellular Pathway
- PMID: 26098895
- PMCID: PMC4476590
- DOI: 10.1371/journal.pone.0131059
Microglia Activate Migration of Glioma Cells through a Pyk2 Intracellular Pathway
Abstract
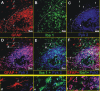
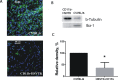
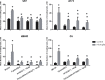
Glioblastoma is one of the most aggressive and fatal brain cancers due to the highly invasive nature of glioma cells. Microglia infiltrate most glioma tumors and, therefore, make up an important component of the glioma microenvironment. In the tumor environment, microglia release factors that lead to the degradation of the extracellular matrix and stimulate signaling pathways to promote glioma cell invasion. In the present study, we demonstrated that microglia can promote glioma migration through a mechanism independent of extracellular matrix degradation. Using western blot analysis, we found upregulation of proline rich tyrosine kinase 2 (Pyk2) protein phosphorylated at Tyr579/580 in glioma cells treated with microglia conditioned medium. This upregulation occurred in rodent C6 and GL261 as well as in human glioma cell lines with varying levels of invasiveness (U-87MG, A172, and HS683). siRNA knock-down of Pyk2 protein and pharmacological blockade by the Pyk2/focal-adhesion kinase (FAK) inhibitor PF-562,271 reversed the stimulatory effect of microglia on glioma migration in all cell lines. A lower concentration of PF-562,271 that selectively inhibits FAK, but not Pyk2, did not have any effect on glioma cell migration. Moreover, with the use of the CD11b-HSVTK microglia ablation mouse model we demonstrated that elimination of microglia in the implanted tumors (GL261 glioma cells were used for brain implantation) by the local in-tumor administration of Ganciclovir, significantly reduced the phosphorylation of Pyk2 at Tyr579/580 in implanted tumor cells. Taken together, these data indicate that microglial cells activate glioma cell migration/dispersal through the pro-migratory Pyk2 signaling pathway in glioma cells.
Conflict of interest statement
Figures
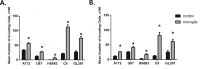



References
-
- Laws ER Jr, Goldberg WJ, Bernsteins JJ. Migration of human malignant astrocytoma cells in the mammalian brain: Scherer revisited. Int J Dev Neurosci. 1993; 11: 691–7. - PubMed
-
- Platten M, Kretz A, Naumann U, Aulwurm S, Egashira K, Isenmann S, et al. Monocyte chemoattractant protein-1 increases microglial infiltration and aggressiveness of gliomas. Ann Neurol. 2003; 54: 388–92. - PubMed
-
- Graeber MB, Scheithauer BW, Kreutzberg GW. Microglia in brain tumors. Glia. 2002; 40: 252–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

