L-type Ca²⁺ channel blockade with antihypertensive medication disrupts VTA synaptic plasticity and drug-associated contextual memory
- PMID: 26100537
- PMCID: PMC4689680
- DOI: 10.1038/mp.2015.84
L-type Ca²⁺ channel blockade with antihypertensive medication disrupts VTA synaptic plasticity and drug-associated contextual memory
Abstract
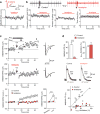
Drug addiction is driven, in part, by powerful and enduring memories of sensory cues associated with drug intake. As such, relapse to drug use during abstinence is frequently triggered by an encounter with drug-associated cues, including the drug itself. L-type Ca(2+) channels (LTCCs) are known to regulate different forms of synaptic plasticity, the major neural substrate for learning and memory, in various brain areas. Long-term potentiation (LTP) of NMDA receptor (NMDAR)-mediated glutamatergic transmission in the ventral tegmental area (VTA) may contribute to the increased motivational valence of drug-associated cues triggering relapse. In this study, using rat brain slices, we found that isradipine, a general LTCC antagonist used as antihypertensive medication, not only blocks the induction of NMDAR LTP but also promotes the reversal of previously induced LTP in the VTA. In behaving rats, isradipine injected into the VTA suppressed the acquisition of cocaine-paired contextual cue memory assessed using a conditioned place preference (CPP) paradigm. Furthermore, administration of isradipine or a CaV1.3 subtype-selective LTCC antagonist (systemic or intra-VTA) before a single extinction or reinstatement session, while having no immediate effect at the time of administration, abolished previously acquired cocaine and alcohol (ethanol) CPP on subsequent days. Notably, CPP thus extinguished cannot be reinstated by drug re-exposure, even after 2 weeks of withdrawal. These results suggest that LTCC blockade during exposure to drug-associated cues may cause unlearning of the increased valence of those cues, presumably via reversal of glutamatergic synaptic plasticity in the VTA.
Conflict of interest statement
Figures
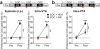



Similar articles
-
Extracellular signal-regulated kinase signaling in the ventral tegmental area mediates cocaine-induced synaptic plasticity and rewarding effects.J Neurosci. 2011 Aug 3;31(31):11244-55. doi: 10.1523/JNEUROSCI.1040-11.2011. J Neurosci. 2011. PMID: 21813685 Free PMC article.
-
Repeated social defeat stress enhances glutamatergic synaptic plasticity in the VTA and cocaine place conditioning.Elife. 2016 Jul 4;5:e15448. doi: 10.7554/eLife.15448. Elife. 2016. PMID: 27374604 Free PMC article.
-
The L-type calcium channel blocker, isradipine, attenuates cue-induced cocaine-seeking by enhancing dopaminergic activity in the ventral tegmental area to nucleus accumbens pathway.Neuropsychopharmacology. 2018 Nov;43(12):2361-2372. doi: 10.1038/s41386-018-0080-2. Epub 2018 May 3. Neuropsychopharmacology. 2018. PMID: 29773910 Free PMC article.
-
L-type calcium channel regulation of dopamine activity in the ventral tegmental area to nucleus accumbens pathway: Implications for substance use, mood disorders and co-morbidities.Neuropharmacology. 2023 Feb 15;224:109336. doi: 10.1016/j.neuropharm.2022.109336. Epub 2022 Nov 19. Neuropharmacology. 2023. PMID: 36414149 Free PMC article. Review.
-
Usage of L-type calcium channel blockers to suppress drug reward and memory driving addiction: Past, present, and future.Neuropharmacology. 2022 Dec 15;221:109290. doi: 10.1016/j.neuropharm.2022.109290. Epub 2022 Oct 12. Neuropharmacology. 2022. PMID: 36241085 Free PMC article. Review.
Cited by
-
Pleiotropic Association of CACNA1C Variants With Neuropsychiatric Disorders.Schizophr Bull. 2023 Sep 7;49(5):1174-1184. doi: 10.1093/schbul/sbad073. Schizophr Bull. 2023. PMID: 37306960 Free PMC article.
-
Cocaine- and stress-primed reinstatement of drug-associated memories elicit differential behavioral and frontostriatal circuit activity patterns via recruitment of L-type Ca2+ channels.Mol Psychiatry. 2020 Oct;25(10):2373-2391. doi: 10.1038/s41380-019-0513-2. Epub 2019 Sep 9. Mol Psychiatry. 2020. PMID: 31501511 Free PMC article.
-
Applications of isradipine in human addiction studies: A systematic literature review.Exp Clin Psychopharmacol. 2023 Apr;31(2):507-522. doi: 10.1037/pha0000633. Epub 2023 Jan 2. Exp Clin Psychopharmacol. 2023. PMID: 36595455 Free PMC article.
-
Ca2+ channel blockade reduces cocaine's vasoconstriction and neurotoxicity in the prefrontal cortex.Transl Psychiatry. 2021 Sep 6;11(1):459. doi: 10.1038/s41398-021-01573-7. Transl Psychiatry. 2021. PMID: 34489397 Free PMC article.
-
Extinction of Contextual Cocaine Memories Requires Cav1.2 within D1R-Expressing Cells and Recruits Hippocampal Cav1.2-Dependent Signaling Mechanisms.J Neurosci. 2017 Dec 6;37(49):11894-11911. doi: 10.1523/JNEUROSCI.2397-17.2017. Epub 2017 Oct 31. J Neurosci. 2017. PMID: 29089442 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

