Aquaporin-3-mediated hydrogen peroxide transport is required for NF-κB signalling in keratinocytes and development of psoriasis
- PMID: 26100668
- PMCID: PMC5628617
- DOI: 10.1038/ncomms8454
Aquaporin-3-mediated hydrogen peroxide transport is required for NF-κB signalling in keratinocytes and development of psoriasis
Abstract
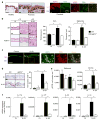
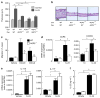
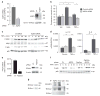
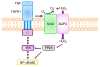
Aquaporin 3 (AQP3), a water/glycerol channel protein, has been found to transport hydrogen peroxide (H2O2). Here, we show that H2O2, imported via AQP3, is involved in nuclear factor-κB (NF-κB) signalling in keratinocytes and in the pathogenesis of psoriasis. IL-23-mediated induction of psoriasis is reduced in AQP3 knockout mice (AQP3(-/-)), and is accompanied by impaired NF-κB activation and intracellular H2O2 accumulation. In primary keratinocyte cultures, cellular import of H2O2 produced by membrane NADPH oxidase 2 (Nox2) in response to TNF-α is facilitated by AQP3 and required for NF-κB activation by regulation of protein phosphatase 2A. As AQP3 associates with Nox2, we propose that this interplay constitutes H2O2-mediated signalling in response to TNF-α stimulation. Collectively, these data indicate that AQP3-facilitated H2O2 transport is required for NF-κB activation in keratinocytes in the development of psoriasis.
Conflict of interest statement
Figures





Similar articles
-
Involvement of aquaporin-3 in epidermal growth factor receptor signaling via hydrogen peroxide transport in cancer cells.Biochem Biophys Res Commun. 2016 Mar 18;471(4):603-9. doi: 10.1016/j.bbrc.2016.02.010. Epub 2016 Feb 16. Biochem Biophys Res Commun. 2016. PMID: 26896765
-
Inhibition of aquaporin-3 in macrophages by a monoclonal antibody as potential therapy for liver injury.Nat Commun. 2020 Nov 9;11(1):5666. doi: 10.1038/s41467-020-19491-5. Nat Commun. 2020. PMID: 33168815 Free PMC article.
-
Aquaporin-3 Controls Breast Cancer Cell Migration by Regulating Hydrogen Peroxide Transport and Its Downstream Cell Signaling.Mol Cell Biol. 2016 Feb 1;36(7):1206-18. doi: 10.1128/MCB.00971-15. Mol Cell Biol. 2016. PMID: 26830227 Free PMC article.
-
Aquaporin-3 in the epidermis: more than skin deep.Am J Physiol Cell Physiol. 2020 Jun 1;318(6):C1144-C1153. doi: 10.1152/ajpcell.00075.2020. Epub 2020 Apr 8. Am J Physiol Cell Physiol. 2020. PMID: 32267715 Free PMC article. Review.
-
Role of hydrogen peroxide in NF-kappaB activation: from inducer to modulator.Antioxid Redox Signal. 2009 Sep;11(9):2223-43. doi: 10.1089/ars.2009.2601. Antioxid Redox Signal. 2009. PMID: 19496701 Review.
Cited by
-
Classification and Gene Structure of Aquaporins.Adv Exp Med Biol. 2023;1398:1-13. doi: 10.1007/978-981-19-7415-1_1. Adv Exp Med Biol. 2023. PMID: 36717483
-
MiR-181b modulates EGFR-dependent VCAM-1 expression and monocyte adhesion in glioblastoma.Oncogene. 2017 Aug 31;36(35):5006-5022. doi: 10.1038/onc.2017.129. Epub 2017 May 1. Oncogene. 2017. PMID: 28459461
-
Therapeutic benefits of glycerol in dry eye disease.Front Med (Lausanne). 2025 Jan 15;11:1531670. doi: 10.3389/fmed.2024.1531670. eCollection 2024. Front Med (Lausanne). 2025. PMID: 39882517 Free PMC article.
-
Identification of potential biomarkers and therapeutic targets in psoriasis based on NF-κB pathway-related genes.Arch Dermatol Res. 2025 Mar 22;317(1):615. doi: 10.1007/s00403-025-04017-0. Arch Dermatol Res. 2025. PMID: 40119934
-
AQP3-mediated H2 O2 uptake inhibits LUAD autophagy by inactivating PTEN.Cancer Sci. 2021 Aug;112(8):3278-3292. doi: 10.1111/cas.15008. Epub 2021 Jun 27. Cancer Sci. 2021. PMID: 34091997 Free PMC article.
References
-
- Perera GK, Di Meglio P, Nestle FO. Psoriasis. Annu Rev Pathol. 2012;7:385–422. - PubMed
-
- Elder JT, et al. Molecular dissection of psoriasis: integrating genetics and biology. J Invest Dermatol. 2010;130:1213–1226. - PubMed
-
- Oka A, Mabuchi T, Ozawa A, Inoko H. Current understanding of human genetics and genetic analysis of psoriasis. J Dermatol. 2012;39:231–241. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

