An in vitro combined antibiotic-antibody treatment eliminates toxicity from Shiga toxin-producing Escherichia coli
- PMID: 26100707
- PMCID: PMC4538565
- DOI: 10.1128/AAC.00763-15
An in vitro combined antibiotic-antibody treatment eliminates toxicity from Shiga toxin-producing Escherichia coli
Abstract
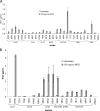
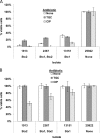
Treating Shiga toxin-producing Escherichia coli (STEC) gastrointestinal infections is difficult. The utility of antibiotics for STEC treatment is controversial, since antibiotic resistance among STEC isolates is widespread and certain antibiotics dramatically increase the expression of Shiga toxins (Stxs), which are some of the most important virulence factors in STEC. Stxs contribute to life-threatening hemolytic uremic syndrome (HUS), which develops in considerable proportions of patients with STEC infections. Understanding the antibiotic resistance profiles of STEC isolates and the Stx induction potential of promising antibiotics is essential for evaluating any antibiotic treatment of STEC. In this study, 42 O157:H7 or non-O157 STEC isolates (including the "big six" serotypes) were evaluated for their resistance against 22 antibiotics by using an antibiotic array. Tigecycline inhibited the growth of all of the tested STEC isolates and also inhibited the production of Stxs (Stx2 in particular). In combination with neutralizing antibodies to Stx1 and Stx2, the tigecycline-antibody treatment fully protected Vero cells from Stx toxicity, even when the STEC bacteria and the Vero cells were cultured together. The combination of an antibiotic such as tigecycline with neutralizing antibodies presents a promising strategy for future STEC treatments.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures



Similar articles
-
Effect of antibiotics on cellular stress generated in Shiga toxin-producing Escherichia coli O157:H7 and non-O157 biofilms.Toxicol In Vitro. 2015 Oct;29(7):1692-700. doi: 10.1016/j.tiv.2015.06.025. Epub 2015 Jun 27. Toxicol In Vitro. 2015. PMID: 26130220
-
Effects of ampicillin, gentamicin, and cefotaxime on the release of Shiga toxins from Shiga toxin-producing Escherichia coli isolated during a diarrhea episode in Faisalabad, Pakistan.Foodborne Pathog Dis. 2010 Jan;7(1):85-90. doi: 10.1089/fpd.2009.0311. Foodborne Pathog Dis. 2010. PMID: 19785532
-
Comparison of Escherichia coli Isolates from humans, food, and farm and companion animals for presence of Shiga toxin-producing E. coli virulence markers.Foodborne Pathog Dis. 2004 Fall;1(3):178-84. doi: 10.1089/fpd.2004.1.178. Foodborne Pathog Dis. 2004. PMID: 15992278
-
New Therapeutic Developments against Shiga Toxin-Producing Escherichia coli.Microbiol Spectr. 2014 Oct;2(5). doi: 10.1128/microbiolspec.EHEC-0013-2013. Microbiol Spectr. 2014. PMID: 26104346 Review.
-
Pathogenesis of Shiga-toxin producing escherichia coli.Curr Top Microbiol Immunol. 2012;357:67-103. doi: 10.1007/82_2011_176. Curr Top Microbiol Immunol. 2012. PMID: 21915773 Review.
Cited by
-
Treatment Strategies for Infections With Shiga Toxin-Producing Escherichia coli.Front Cell Infect Microbiol. 2020 May 6;10:169. doi: 10.3389/fcimb.2020.00169. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 32435624 Free PMC article. Review.
-
Potentiation of Antibiotics by a Novel Antimicrobial Peptide against Shiga Toxin Producing E. coli O157:H7.Sci Rep. 2020 Jun 22;10(1):10029. doi: 10.1038/s41598-020-66571-z. Sci Rep. 2020. PMID: 32572054 Free PMC article.
-
Therapeutic Antibodies Against Shiga Toxins: Trends and Perspectives.Front Cell Infect Microbiol. 2022 Feb 10;12:825856. doi: 10.3389/fcimb.2022.825856. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35223548 Free PMC article. Review.
-
Risk of Hemolytic Uremic Syndrome Related to Treatment of Escherichia coli O157 Infection with Different Antimicrobial Classes.Microorganisms. 2021 Sep 21;9(9):1997. doi: 10.3390/microorganisms9091997. Microorganisms. 2021. PMID: 34576892 Free PMC article.
-
A novel Shiga toxin 2a neutralizing antibody therapeutic with low immunogenicity and high efficacy.Antimicrob Agents Chemother. 2024 Jan 10;68(1):e0059823. doi: 10.1128/aac.00598-23. Epub 2023 Dec 4. Antimicrob Agents Chemother. 2024. PMID: 38047751 Free PMC article.
References
-
- Gould LH, Bopp C, Strockbine N, Atkinson R, Baselski V, Body B, Carey R, Crandall C, Hurd S, Kaplan R, Neill M, Shea S, Somsel P, Tobin-D'Angelo M, Griffin PM, Gerner-Smidt P. 2009. Recommendations for diagnosis of Shiga toxin-producing Escherichia coli infections by clinical laboratories. MMWR Recomm Rep 58(RR-12):1–14. - PubMed
-
- Trofa AF, Ueno-Olsen H, Oiwa R, Yoshikawa M. 1999. Dr. Kiyoshi Shiga: discoverer of the dysentery bacillus. Clin Infect Dis 29:1303–1306. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

