Paternal allelic mutation at the Kcnq1 locus reduces pancreatic β-cell mass by epigenetic modification of Cdkn1c
- PMID: 26100882
- PMCID: PMC4500236
- DOI: 10.1073/pnas.1422104112
Paternal allelic mutation at the Kcnq1 locus reduces pancreatic β-cell mass by epigenetic modification of Cdkn1c
Abstract
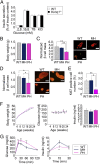
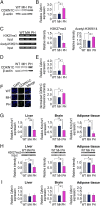
Genetic factors are important determinants of the onset and progression of diabetes mellitus. Numerous susceptibility genes for type 2 diabetes, including potassium voltage-gated channel, KQT-like subfamily Q, member1 (KCNQ1), have been identified in humans by genome-wide analyses and other studies. Experiments with genetically modified mice have also implicated various genes in the pathogenesis of diabetes. However, the possible effects of the parent of origin for diabetes susceptibility alleles on disease onset have remained unclear. Here, we show that a mutation at the Kcnq1 locus reduces pancreatic β-cell mass in mice by epigenetic modulation only when it is inherited from the father. The noncoding RNA KCNQ1 overlapping transcript1 (Kcnq1ot1) is expressed from the Kcnq1 locus and regulates the expression of neighboring genes on the paternal allele. We found that disruption of Kcnq1 results in reduced Kcnq1ot1 expression as well as the increased expression of cyclin-dependent kinase inhibitor 1C (Cdkn1c), an imprinted gene that encodes a cell cycle inhibitor, only when the mutation is on the paternal allele. Furthermore, histone modification at the Cdkn1c promoter region in pancreatic islets was found to contribute to this phenomenon. Our observations suggest that the Kcnq1 genomic region directly regulates pancreatic β-cell mass and that genomic imprinting may be a determinant of the onset of diabetes mellitus.
Keywords: Kcnq1; imprinting; pancreatic β-cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Yamauchi T, et al. A genome-wide association study in the Japanese population identifies susceptibility loci for type 2 diabetes at UBE2E2 and C2CD4A-C2CD4B. Nat Genet. 2010;42(10):864–868. - PubMed
-
- Yasuda K, et al. Variants in KCNQ1 are associated with susceptibility to type 2 diabetes mellitus. Nat Genet. 2008;40(9):1092–1097. - PubMed
-
- Unoki H, et al. SNPs in KCNQ1 are associated with susceptibility to type 2 diabetes in East Asian and European populations. Nat Genet. 2008;40(9):1098–1102. - PubMed
-
- Chen YH, et al. KCNQ1 gain-of-function mutation in familial atrial fibrillation. Science. 2003;299(5604):251–254. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

