Activity-Based Probe for N-Acylethanolamine Acid Amidase
- PMID: 26102511
- PMCID: PMC4575644
- DOI: 10.1021/acschembio.5b00197
Activity-Based Probe for N-Acylethanolamine Acid Amidase
Abstract
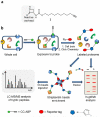
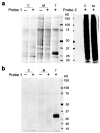
N-Acylethanolamine acid amidase (NAAA) is a lysosomal cysteine hydrolase involved in the degradation of saturated and monounsaturated fatty acid ethanolamides (FAEs), a family of endogenous lipid signaling molecules that includes oleoylethanolamide (OEA) and palmitoylethanolamide (PEA). Among the reported NAAA inhibitors, α-amino-β-lactone (3-aminooxetan-2-one) derivatives have been shown to prevent FAE hydrolysis in innate-immune and neural cells and to reduce reactions to inflammatory stimuli. Recently, we disclosed two potent and selective NAAA inhibitors, the compounds ARN077 (5-phenylpentyl-N-[(2S,3R)-2-methyl-4-oxo-oxetan-3-yl]carbamate) and ARN726 (4-cyclohexylbutyl-N-[(S)-2-oxoazetidin-3-yl]carbamate). The former is active in vivo by topical administration in rodent models of hyperalgesia and allodynia, while the latter exerts systemic anti-inflammatory effects in mouse models of lung inflammation. In the present study, we designed and validated a derivative of ARN726 as the first activity-based protein profiling (ABPP) probe for the in vivo detection of NAAA. The newly synthesized molecule 1 is an effective in vitro and in vivo click-chemistry activity based probe (ABP), which is able to capture the catalytically active form of NAAA in Human Embryonic Kidney 293 (HEK293) cells overexpressing human NAAA as well as in rat lung tissue. Competitive ABPP with 1 confirmed that ARN726 and ARN077 inhibit NAAA in vitro and in vivo. Compound 1 is a useful new tool to identify activated NAAA both in vitro and in vivo and to investigate the physiological and pathological roles of this enzyme.
Figures

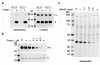

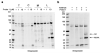
References
-
- Tsuboi K, Sun YX, Okamoto Y, Araki N, Tonai T, Ueda N. Molecular characterization of N-acylethanolamine-hydrolyzing acid amidase, a novel member of the choloylglycine hydrolase family with structural and functional similarity to acid ceramidase. J. Biol. Chem. 2005;280:11082–11092. - PubMed
-
- Tsuboi K, Takezaki N, Ueda N. The N-acylethanolamine-hydrolyzing acid amidase (NAAA) Chem. Biodivers. 2007;4:1914–1925. - PubMed
-
- Ueda N, Tsuboi K, Uyama T. N-acylethanolamine metabolism with special reference to N-acylethanolamine-hydrolyzing acid amidase (NAAA) Prog. Lipid. Res. 2010;49:299–315. - PubMed
-
- Rossocha M, Schultz-Heienbrok R, von Moeller H, Coleman JP, Saenger W. Conjugated bile acid hydrolase is a tetrameric N-terminal thiol hydrolase with specific recognition of its cholyl but not of its tauryl product. Biochemistry. 2005;44:5739–5748. - PubMed
-
- Zhao LY, Tsuboi K, Okamoto Y, Nagahata S, Ueda N. Proteolytic activation and glycosylation of N-acylethanolamine-hydrolyzing acid amidase, a lysosomal enzyme involved in the endocannabinoid metabolism. Biochim. Biophys. Acta. 2007;1771:1397–1405. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

