Metallobiology of Tuberculosis
- PMID: 26103977
- PMCID: PMC5180607
- DOI: 10.1128/microbiolspec.MGM2-0012-2013
Metallobiology of Tuberculosis
Abstract
Transition metals are essential constituents of all living organisms, playing crucial structural and catalytic parts in many enzymes and transcription factors. However, transition metals can also be toxic when present in excess. Their uptake and efflux rates must therefore be carefully controlled by biological systems. In this chapter, we summarize the current knowledge about uptake and efflux systems in Mycobacterium tuberculosis for mainly three of these metals, namely iron, zinc, and copper. We also propose questions for future research in the field of metallobiology of host-pathogen interactions in tuberculosis.
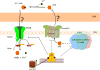
Figures

References
-
- Andrews SC, Robinson AK, Rodriguez-Quinones F. Bacterial iron homeostasis. FEMS microbiology reviews. 2003;27:215–237. - PubMed
-
- Weinberg ED. Iron and susceptibility to infectious disease. Science. 1974;184:952–956. - PubMed
-
- Dobryszycka W. Biological functions of haptoglobin–new pieces to an old puzzle. European journal of clinical chemistry and clinical biochemistry: journal of the Forum of European Clinical Chemistry Societies. 1997;35:647–654. - PubMed
-
- Tolosano E, Altruda F. Hemopexin: structure, function, and regulation. DNA and cell biology. 2002;21:297–306. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

