Near-infrared fluorescent proteins engineered from bacterial phytochromes
- PMID: 26115447
- PMCID: PMC4553112
- DOI: 10.1016/j.cbpa.2015.06.005
Near-infrared fluorescent proteins engineered from bacterial phytochromes
Abstract
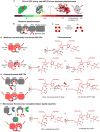
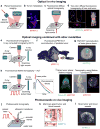
Near-infrared fluorescent proteins (NIR FPs), photoactivatable NIR FPs and NIR reporters of protein-protein interactions developed from bacterial phytochrome photoreceptors (BphPs) have advanced non-invasive deep-tissue imaging. Here we provide a brief guide to the BphP-derived NIR probes with an emphasis on their in vivo applications. We describe phenotypes of NIR FPs and their photochemical and intracellular properties. We discuss NIR FP applications for imaging of various cell types, tissues and animal models in basic and translational research. In this discussion, we focus on NIR FPs that efficiently incorporate endogenous biliverdin chromophore and therefore can be used as straightforward as GFP-like proteins. We also overview a usage of NIR FPs in different imaging platforms, from planar epifluorescence to tomographic and photoacoustic technologies.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures

References
-
- Weissleder R. A clearer vision for in vivo imaging. Nat Biotechnol. 2001;19:316–317. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

