The KCNE2 K⁺ channel regulatory subunit: Ubiquitous influence, complex pathobiology
- PMID: 26123744
- PMCID: PMC4917011
- DOI: 10.1016/j.gene.2015.06.061
The KCNE2 K⁺ channel regulatory subunit: Ubiquitous influence, complex pathobiology
Abstract
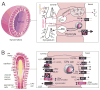
The KCNE single-span transmembrane subunits are encoded by five-member gene families in the human and mouse genomes. Primarily recognized for co-assembling with and functionally regulating the voltage-gated potassium channels, the broad influence of KCNE subunits in mammalian physiology belies their small size. KCNE2 has been widely studied since we first discovered one of its roles in the heart and its association with inherited and acquired human Long QT syndrome. Since then, physiological analyses together with human and mouse genetics studies have uncovered a startling array of functions for KCNE2, in the heart, stomach, thyroid and choroid plexus. The other side of this coin is the variety of interconnected disease manifestations caused by KCNE2 disruption, involving both excitable cells such as cardiomyocytes, and non-excitable, polarized epithelia. Kcne2 deletion in mice has been particularly instrumental in illustrating the potential ramifications within a monogenic arrhythmia syndrome, with removal of one piece revealing the unexpected complexity of the puzzle. Here, we review current knowledge of the function and pathobiology of KCNE2.
Keywords: Cardiac arrhythmia; Potassium channel; Sudden cardiac death; Thyroid; Transporter.
Copyright © 2015 Elsevier B.V. All rights reserved.
Figures





References
-
- Catterall WA. The molecular basis of neuronal excitability. Science. 1984;223(4637):653–61. - PubMed
-
- Hille B, Armstrong CM, MacKinnon R. Ion channels: from idea to reality. Nature medicine. 1999;5(10):1105–9. - PubMed
-
- Abbott GW. Biology of the KCNQ1 potassium channel. New Journal of Science. 2014
-
- MacKinnon R. Determination of the subunit stoichiometry of a voltage-activated potassium channel. Nature. 1991;350(6315):232–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

