Phase II Trial of Goserelin and Exemestane Combination Therapy in Premenopausal Women With Locally Advanced or Metastatic Breast Cancer
- PMID: 26131799
- PMCID: PMC4504532
- DOI: 10.1097/MD.0000000000001006
Phase II Trial of Goserelin and Exemestane Combination Therapy in Premenopausal Women With Locally Advanced or Metastatic Breast Cancer
Abstract
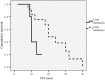
A promising option as the treatment of choice for premenopausal patients with locally advanced or metastatic breast cancer (MBC) could be the combination of a luteinizing hormone-releasing hormone analog and an aromatase inhibitor. However, no prospective studies on the efficacy of goserelin with exemestane in locally advanced or MBC premenopausal breast cancer patients have been reported.We present the phase II trial of goserelin plus exemestane in a total of 44 premenopausal women with locally advanced or MBC. All patients received a subcutaneous injection of 3.6 mg goserelin every 4 weeks along with 25 mg exemestane daily. The primary end point was progression-free survival (PFS). The second end point included overall survival (OS), objective response rate (ORR), duration of response (DOR), and clinical benefit rate (CBR) based on complete response (CR), partial response (PR), or stable disease (SD) for ≥6 months.The median PFS was 13 months (range: 2-42 months). The median DOR was 8 months (range: 2-40 months). Two patients achieved CR (4.5%), and 15 patients experienced PR (34.1%). Fifteen patients (34.1%) had SD ≥6 months. The ORR was 38.6%, and the CBR was 65.9%. Primary progressive disease occurred in 15 patients (34.1%). Five patients (11.4%) died during the study period. Because a few patients have died, the median OS has not been reached. Drug therapy was well tolerated. The most frequent grade-3 adverse events were arthralgia (18.2%), skin rash (6.8%), and myalgia (4.5%). No participants withdrew from the study due to drug toxicity.This study suggested that goserelin and exemestane might be highly effective and well-tolerated regimens in premenopausal women with hormone-responsive, locally advanced or MBC.
Conflict of interest statement
The authors have no funding and conflicts of interest to disclose.
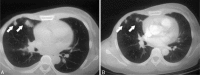
Figures
Similar articles
-
Goserelin plus letrozole as first- or second-line hormonal treatment in premenopausal patients with advanced breast cancer.Endocr J. 2011;58(6):509-16. doi: 10.1507/endocrj.k11e-020. Epub 2011 Apr 29. Endocr J. 2011. PMID: 21532215
-
[Goserelin plus anastrozole in the treatment of premenopausal patients with metastatic breast cancer].Zhonghua Yi Xue Za Zhi. 2010 Mar 2;90(8):526-8. Zhonghua Yi Xue Za Zhi. 2010. PMID: 20367963 Chinese.
-
Fulvestrant plus goserelin versus anastrozole plus goserelin versus goserelin alone for hormone receptor-positive, HER2-negative tamoxifen-pretreated premenopausal women with recurrent or metastatic breast cancer (KCSG BR10-04): a multicentre, open-label, three-arm, randomised phase II trial (FLAG study).Eur J Cancer. 2018 Nov;103:127-136. doi: 10.1016/j.ejca.2018.08.004. Epub 2018 Sep 14. Eur J Cancer. 2018. PMID: 30223226 Clinical Trial.
-
Complete estrogen blockade for the treatment of metastatic and early stage breast cancer.Drugs Aging. 2000 Apr;16(4):261-71. doi: 10.2165/00002512-200016040-00002. Drugs Aging. 2000. PMID: 10874521 Review.
-
Use of goserelin in the treatment of breast cancer.Expert Rev Anticancer Ther. 2005 Aug;5(4):591-604. doi: 10.1586/14737140.5.4.591. Expert Rev Anticancer Ther. 2005. PMID: 16111461 Review.
Cited by
-
Pharmacotherapeutic Targeting of G Protein-Coupled Receptors in Oncology: Examples of Approved Therapies and Emerging Concepts.Drugs. 2017 Jun;77(9):951-965. doi: 10.1007/s40265-017-0738-9. Drugs. 2017. PMID: 28401445 Review.
-
Bilateral Salpingo-oophorectomy Compared to Gonadotropin-Releasing Hormone Agonists in Premenopausal Hormone Receptor-Positive Metastatic Breast Cancer Patients Treated with Aromatase Inhibitors.Cancer Res Treat. 2017 Oct;49(4):1153-1163. doi: 10.4143/crt.2016.463. Epub 2017 Feb 27. Cancer Res Treat. 2017. PMID: 28253566 Free PMC article.
-
Five-Fraction High-Conformal Ultrahypofractionated Radiotherapy for Primary Tumors in Metastatic Breast Cancer.J Breast Cancer. 2024 Apr;27(2):91-104. doi: 10.4048/jbc.2024.0004. Epub 2024 Mar 26. J Breast Cancer. 2024. PMID: 38529591 Free PMC article.
-
Palbociclib Combined with Fulvestrant in Premenopausal Women with Advanced Breast Cancer and Prior Progression on Endocrine Therapy: PALOMA-3 Results.Oncologist. 2017 Sep;22(9):1028-1038. doi: 10.1634/theoncologist.2017-0072. Epub 2017 Jun 26. Oncologist. 2017. PMID: 28652278 Free PMC article. Clinical Trial.
-
Goserelin-Induced Chemical Burn: A Case Report and Review of the Literature.Cureus. 2023 Sep 21;15(9):e45692. doi: 10.7759/cureus.45692. eCollection 2023 Sep. Cureus. 2023. PMID: 37745751 Free PMC article.
References
-
- Coughlin SS, Ekwueme DU. Breast cancer as a global health concern. Cancer Epidemiol 2009; 33:315–318. - PubMed
-
- Yuan JM, Yu MC, Ross RK, et al. Risk factors for breast cancer in Chinese women in Shanghai. Cancer Res 1988; 48:1949–1953. - PubMed
-
- Wang QS, Ross RK, Yu MC, et al. A case-control study of breast cancer in Tianjin, China. Cancer Epidemiol 1992; 1:435–439. - PubMed
-
- Yang L, Parkin DM, Li L, et al. Time trends in cancer mortality in China: 1987–1999. Int J Cancer 2003; 106:771–783. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

