Differential thermosensitivity in mixed syndrome cardiac sodium channel mutants
- PMID: 26131924
- PMCID: PMC4594293
- DOI: 10.1113/JP270139
Differential thermosensitivity in mixed syndrome cardiac sodium channel mutants
Abstract
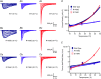
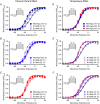
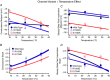
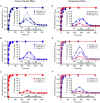
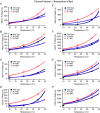
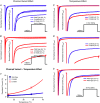
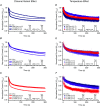
Cardiac arrhythmias are often associated with mutations in SCN5A the gene that encodes the cardiac paralogue of the voltage-gated sodium channel, NaV 1.5. The NaV 1.5 mutants R1193Q and E1784K give rise to both long QT and Brugada syndromes. Various environmental factors, including temperature, may unmask arrhythmia. We sought to determine whether temperature might be an arrhythmogenic trigger in these two mixed syndrome mutants. Whole-cell patch clamp was used to measure the biophysical properties of NaV 1.5 WT, E1784K and R1193Q mutants. Recordings were performed using Chinese hamster ovary (CHOk1) cells transiently transfected with the NaV 1.5 α subunit (WT, E1784K, or R1193Q), β1 subunit, and eGFP. The channels' voltage-dependent and kinetic properties were measured at three different temperatures: 10ºC, 22ºC, and 34ºC. The E1784K mutant is more thermosensitive than either WT or R1193Q channels. When temperature is elevated from 22°C to 34°C, there is a greater increase in late INa and use-dependent inactivation in E1784K than in WT or R1193Q. However, when temperature is lowered to 10°C, the two mutants show a decrease in channel availability. Action potential modelling using Q10 fit values, extrapolated to physiological and febrile temperatures, show a larger transmural voltage gradient in E1784K compared to R1193Q and WT with hyperthermia. The E1784K mutant is more thermosensitive than WT or R1193Q channels. This enhanced thermosensitivity may be a mechanism for arrhythmogenesis in patients with E1784K sodium channels.
© 2015 The Authors. The Journal of Physiology © 2015 The Physiological Society.
Figures




References
-
- Antzelevitch C, Brugada P, Borggrefe M, Brugada J, Brugada R, Corrado D, Gussak I, LeMarec H, Nademanee K, Perez Riera AR, et al. Brugada syndrome: Report of the second consensus conference: Endorsed by the Heart Rhythm Society and the Chinese Heart Rhythm Association. Circulation. 2005;111:659–670. - PubMed
-
- Bezzina C, Veldkamp MW, van Den Berg MP, Postma AV, Rook MB, Viersma JW, van Langen IM, Tan-Sindhunata G, Bink-Boelkens MT, van Der Hout AH, et al. A single Na+ channel mutation causing both long-QT and Brugada syndromes. Circ Res. 1999;85:1206–1213. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

