Eriodictyol Protects Endothelial Cells against Oxidative Stress-Induced Cell Death through Modulating ERK/Nrf2/ARE-Dependent Heme Oxygenase-1 Expression
- PMID: 26132561
- PMCID: PMC4519856
- DOI: 10.3390/ijms160714526
Eriodictyol Protects Endothelial Cells against Oxidative Stress-Induced Cell Death through Modulating ERK/Nrf2/ARE-Dependent Heme Oxygenase-1 Expression
Abstract
The pathophysiology of cardiovascular diseases is complex and may involve oxidative stress-related pathways. Eriodictyol is a flavonoid present in citrus fruits that demonstrates anti-inflammatory, anti-cancer, neurotrophic, and antioxidant effects in a range of pathophysiological conditions including vascular diseases. Because oxidative stress plays a key role in the pathogenesis of cardiovascular disease, the present study was designed to verify whether eriodictyol has therapeutic potential. Upregulation of heme oxygenase-1 (HO-1), a phase II detoxifying enzyme, in endothelial cells is considered to be helpful in cardiovascular disease. In this study, human umbilical vein endothelial cells (HUVECs) treated with eriodictyol showed the upregulation of HO-1 through extracellular-regulated kinase (ERK)/nuclear factor erythroid 2-related factor 2 (Nrf2)/antioxidant response element (ARE) signaling pathways. Further, eriodictyol treatment provided protection against hydrogen peroxide-provoked cell death. This protective effect was eliminated by treatment with a specific inhibitor of HO-1 and RNA interference-mediated knockdown of HO-1 expression. These data demonstrate that eriodictyol induces ERK/Nrf2/ARE-mediated HO-1 upregulation in human endothelial cells, which is directly associated with its vascular protection against oxidative stress-related endothelial injury, and propose that targeting the upregulation of HO-1 is a promising approach for therapeutic intervention in cardiovascular disease.
Keywords: cell death; endothelial cells; eriodictyol; flavonoid; heme oxygenase-1; oxidative stress.
Figures
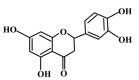
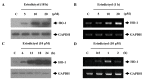

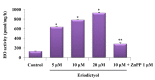
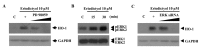
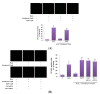
 , dose increasing.
, dose increasing.
 , dose increasing.
, dose increasing.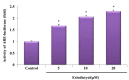
Similar articles
-
Epigallocatechin-3-gallate protects HUVECs from PM2.5-induced oxidative stress injury by activating critical antioxidant pathways.Molecules. 2015 Apr 14;20(4):6626-39. doi: 10.3390/molecules20046626. Molecules. 2015. PMID: 25875041 Free PMC article.
-
Miltirone protects human EA.hy926 endothelial cells from oxidized low-density lipoprotein-derived oxidative stress via a heme oxygenase-1 and MAPK/Nrf2 dependent pathway.Phytomedicine. 2016 Dec 15;23(14):1806-1813. doi: 10.1016/j.phymed.2016.11.003. Epub 2016 Nov 4. Phytomedicine. 2016. PMID: 27912883
-
Fisetin induces Nrf2-mediated HO-1 expression through PKC-δ and p38 in human umbilical vein endothelial cells.J Cell Biochem. 2011 Sep;112(9):2352-60. doi: 10.1002/jcb.23158. J Cell Biochem. 2011. PMID: 21520244
-
Activation of Nrf2/HO-1 signaling: An important molecular mechanism of herbal medicine in the treatment of atherosclerosis via the protection of vascular endothelial cells from oxidative stress.J Adv Res. 2021 Jul 6;34:43-63. doi: 10.1016/j.jare.2021.06.023. eCollection 2021 Dec. J Adv Res. 2021. PMID: 35024180 Free PMC article. Review.
-
Synergistic Interaction Between Heme Oxygenase (HO) and Nuclear-Factor E2- Related Factor-2 (Nrf2) against Oxidative Stress in Cardiovascular Related Diseases.Curr Pharm Des. 2017;23(10):1465-1470. doi: 10.2174/1381612823666170113153818. Curr Pharm Des. 2017. PMID: 28088909 Review.
Cited by
-
Activation of protein kinase C-α/heme oxygenase-1 signaling pathway improves mitochondrial dynamics in lipopolysaccharide-activated NR8383 cells.Exp Ther Med. 2018 Aug;16(2):1529-1537. doi: 10.3892/etm.2018.6290. Epub 2018 Jun 12. Exp Ther Med. 2018. PMID: 30112072 Free PMC article.
-
Erythronium japonicum Alleviates Inflammatory Pain by Inhibiting MAPK Activation and by Suppressing NF-κB Activation via ERK/Nrf2/HO-1 Signaling Pathway.Antioxidants (Basel). 2020 Jul 16;9(7):626. doi: 10.3390/antiox9070626. Antioxidants (Basel). 2020. PMID: 32708683 Free PMC article.
-
Role of SLC7A11/xCT in Ovarian Cancer.Int J Mol Sci. 2024 Jan 2;25(1):587. doi: 10.3390/ijms25010587. Int J Mol Sci. 2024. PMID: 38203758 Free PMC article. Review.
-
Natural flavonoids from herbs and nutraceuticals as ferroptosis inhibitors in central nervous system diseases: current preclinical evidence and future perspectives.Front Pharmacol. 2025 Mar 24;16:1570069. doi: 10.3389/fphar.2025.1570069. eCollection 2025. Front Pharmacol. 2025. PMID: 40196367 Free PMC article. Review.
-
Chemical studies on the parasitic plant Thonningia sanguinea Vahl.RSC Adv. 2018 Jun 7;8(37):21002-21011. doi: 10.1039/c8ra03913e. eCollection 2018 Jun 5. RSC Adv. 2018. PMID: 35542344 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

