miR-16 rescues F508del-CFTR function in native cystic fibrosis epithelial cells
- PMID: 26133785
- PMCID: PMC5488273
- DOI: 10.1038/gt.2015.56
miR-16 rescues F508del-CFTR function in native cystic fibrosis epithelial cells
Abstract
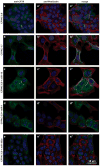
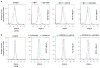
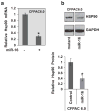
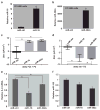
Cystic fibrosis (CF) is due to mutations in the CFTR gene, which prevents correct folding, trafficking and function of the mutant cystic fibrosis transmembrane conductance regulator (CFTR) protein. The dysfunctional effect of CFTR mutations, principally the F508del-CFTR mutant, is further manifested by hypersecretion of the pro-inflammatory chemokine interleukin-8 into the airway lumen, which further contributes to morbidity and mortality. We have hypothesized that microRNA (miR)-based therapeutics could rescue the dysfunctional consequences of mutant CFTR. Here we report that a miR-16 mimic can effectively rescue F508del-CFTR protein function in airway cell lines and primary cultures, of differentiated human bronchial epithelia from F508del homozygotes, which express mutant CFTR endogenously. We also identify two other miRs, miR-1 and miR-302a, which are also active. Although miR-16 is expressed at basal comparable levels in CF and control cells, miR-1 and miR-302a are undetectable. When miR mimics are expressed in CF lung or pancreatic cells, the expression of the F508del-CFTR protein is significantly increased. Importantly, miR-16 promotes functional rescue of the cyclic AMP-activated apical F508del-CFTR chloride channel in primary lung epithelial cells from CF patients. We interpret these findings to suggest that these miRs may constitute novel targets for CF therapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Reduced expression of Tis7/IFRD1 protein in murine and human cystic fibrosis airway epithelial cell models homozygous for the F508del-CFTR mutation.Biochem Biophys Res Commun. 2011 Aug 5;411(3):471-6. doi: 10.1016/j.bbrc.2011.06.104. Epub 2011 Jun 24. Biochem Biophys Res Commun. 2011. PMID: 21723850
-
Augmentation of Cystic Fibrosis Transmembrane Conductance Regulator Function in Human Bronchial Epithelial Cells via SLC6A14-Dependent Amino Acid Uptake. Implications for Treatment of Cystic Fibrosis.Am J Respir Cell Mol Biol. 2019 Dec;61(6):755-764. doi: 10.1165/rcmb.2019-0094OC. Am J Respir Cell Mol Biol. 2019. PMID: 31189070
-
MicroRNA-145 Antagonism Reverses TGF-β Inhibition of F508del CFTR Correction in Airway Epithelia.Am J Respir Crit Care Med. 2018 Mar 1;197(5):632-643. doi: 10.1164/rccm.201704-0732OC. Am J Respir Crit Care Med. 2018. PMID: 29232160 Free PMC article.
-
Pharmacological Correction of Cystic Fibrosis: Molecular Mechanisms at the Plasma Membrane to Augment Mutant CFTR Function.Curr Drug Targets. 2016;17(11):1275-81. doi: 10.2174/1389450117666151209114343. Curr Drug Targets. 2016. PMID: 26648081 Review.
-
Lumacaftor and ivacaftor in the management of patients with cystic fibrosis: current evidence and future prospects.Ther Adv Respir Dis. 2015 Dec;9(6):313-26. doi: 10.1177/1753465815601934. Epub 2015 Sep 28. Ther Adv Respir Dis. 2015. PMID: 26416827 Review.
Cited by
-
Integrative chemogenomic analysis identifies small molecules that partially rescue ΔF508-CFTR for cystic fibrosis.CPT Pharmacometrics Syst Pharmacol. 2021 May;10(5):500-510. doi: 10.1002/psp4.12626. Epub 2021 May 2. CPT Pharmacometrics Syst Pharmacol. 2021. PMID: 33934548 Free PMC article.
-
Role of Non-Coding RNAs in Post-Transcriptional Regulation of Lung Diseases.Front Genet. 2021 Nov 8;12:767348. doi: 10.3389/fgene.2021.767348. eCollection 2021. Front Genet. 2021. PMID: 34819948 Free PMC article. Review.
-
The role of microRNAs in chronic respiratory disease: recent insights.Biol Chem. 2018 Feb 23;399(3):219-234. doi: 10.1515/hsz-2017-0249. Biol Chem. 2018. PMID: 29148977 Free PMC article. Review.
-
Inhibition of MicroRNA 6937 Delays Photoreceptor and Vision Loss in a Mouse Model of Retinitis Pigmentosa.Pharmaceutics. 2020 Sep 24;12(10):913. doi: 10.3390/pharmaceutics12100913. Pharmaceutics. 2020. PMID: 32987664 Free PMC article.
-
Perspectives in MicroRNA Therapeutics for Cystic Fibrosis.Noncoding RNA. 2025 Jan 12;11(1):3. doi: 10.3390/ncrna11010003. Noncoding RNA. 2025. PMID: 39846681 Free PMC article. Review.
References
-
- Riordan JR, Rommens JM, Kerem B, Alon N, Rozmahel R, Grzelczak Z, et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science. 1989;245:1066–1073. - PubMed
-
- Rommens JM, Iannuzzi MC, Kerem B, Drumm ML, Melmer G, Dean M, et al. Identification of the cystic fibrosis gene: chromosome walking and jumping. Science. 1989;245:1059–1065. - PubMed
-
- Bobadilla JL, Macek M, Jr, Fine JP, Farrell PM. Cystic fibrosis: a worldwide analysis of CFTR mutations–correlation with incidence data and application to screening. Hum Mutat. 2002;19:575–606. - PubMed
-
- Welsh MJ, Denning GM, Ostedgaard LS, Anderson MP. Dysfunction of CFTR bearing the delta F508 mutation. J Cell Sci. 1993;17:235–239. - PubMed
-
- Dean TP, Dai Y, Shute JK, Church MK, Warner JO. Interleukin-8 concentrations are elevated in bronchoalveolar lavage, sputum, and sera of children with cystic fibrosis. Pediatr Res. 1993;34:159–161. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

