Glutamate Stimulates Local Protein Synthesis in the Axons of Rat Cortical Neurons by Activating α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic Acid (AMPA) Receptors and Metabotropic Glutamate Receptors
- PMID: 26134564
- PMCID: PMC4543638
- DOI: 10.1074/jbc.M115.638023
Glutamate Stimulates Local Protein Synthesis in the Axons of Rat Cortical Neurons by Activating α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic Acid (AMPA) Receptors and Metabotropic Glutamate Receptors
Abstract
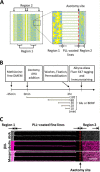
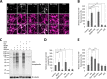
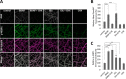
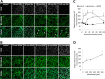
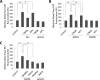
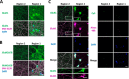
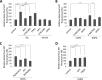
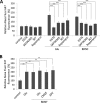
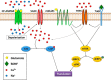
Glutamate is the principal excitatory neurotransmitter in the mammalian CNS. By analyzing the metabolic incorporation of azidohomoalanine, a methionine analogue, in newly synthesized proteins, we find that glutamate treatments up-regulate protein translation not only in intact rat cortical neurons in culture but also in the axons emitting from cortical neurons before making synapses with target cells. The process by which glutamate stimulates local translation in axons begins with the binding of glutamate to the ionotropic AMPA receptors and metabotropic glutamate receptor 1 and members of group 2 metabotropic glutamate receptors on the plasma membrane. Subsequently, the activated mammalian target of rapamycin (mTOR) signaling pathway and the rise in Ca(2+), resulting from Ca(2+) influxes through calcium-permeable AMPA receptors, voltage-gated Ca(2+) channels, and transient receptor potential canonical channels, in axons stimulate the local translation machinery. For comparison, the enhancement effects of brain-derived neurotrophic factor (BDNF) on the local protein synthesis in cortical axons were also studied. The results indicate that Ca(2+) influxes via transient receptor potential canonical channels and activated the mTOR pathway in axons also mediate BDNF stimulation to local protein synthesis. However, glutamate- and BDNF-induced enhancements of translation in axons exhibit different kinetics. Moreover, Ca(2+) and mTOR signaling appear to play roles carrying different weights, respectively, in transducing glutamate- and BDNF-induced enhancements of axonal translation. Thus, our results indicate that exposure to transient increases of glutamate and more lasting increases of BDNF would stimulate local protein synthesis in migrating axons en route to their targets in the developing brain.
Keywords: axon; glutamate; local translation; metabotropic glutamate receptor (mGluR); protein synthesis; α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPA receptor, AMPAR).
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures
Similar articles
-
Brain-derived neurotrophic factor activation of CaM-kinase kinase via transient receptor potential canonical channels induces the translation and synaptic incorporation of GluA1-containing calcium-permeable AMPA receptors.J Neurosci. 2012 Jun 13;32(24):8127-37. doi: 10.1523/JNEUROSCI.6034-11.2012. J Neurosci. 2012. PMID: 22699894 Free PMC article.
-
Glutamate receptors on myelinated spinal cord axons: II. AMPA and GluR5 receptors.Ann Neurol. 2009 Feb;65(2):160-6. doi: 10.1002/ana.21539. Ann Neurol. 2009. PMID: 19224531 Free PMC article.
-
Mature hippocampal astrocytes exhibit functional metabotropic and ionotropic glutamate receptors in situ.Glia. 1999 Mar;26(1):1-11. doi: 10.1002/(sici)1098-1136(199903)26:1<1::aid-glia1>3.0.co;2-z. Glia. 1999. PMID: 10088667
-
Auxiliary subunits assist AMPA-type glutamate receptors.Science. 2006 Mar 3;311(5765):1253-6. doi: 10.1126/science.1123339. Science. 2006. PMID: 16513974 Review.
-
Interacting partners of AMPA-type glutamate receptors.J Mol Neurosci. 2012 Oct;48(2):441-7. doi: 10.1007/s12031-012-9724-6. Epub 2012 Feb 24. J Mol Neurosci. 2012. PMID: 22361832 Review.
Cited by
-
Expanding Axonal Transcriptome Brings New Functions for Axonally Synthesized Proteins in Health and Disease.Neuroscientist. 2018 Apr;24(2):111-129. doi: 10.1177/1073858417712668. Epub 2017 Jun 8. Neuroscientist. 2018. PMID: 28593814 Free PMC article. Review.
-
Acute Manganese Exposure Modifies the Translation Machinery via PI3K/Akt Signaling in Glial Cells.ASN Neuro. 2022 Jan-Dec;14:17590914221131452. doi: 10.1177/17590914221131452. ASN Neuro. 2022. PMID: 36203371 Free PMC article.
-
mTOR kinase activity disrupts a phosphorylation signaling network in schizophrenia brain.Mol Psychiatry. 2021 Nov;26(11):6868-6879. doi: 10.1038/s41380-021-01135-9. Epub 2021 May 14. Mol Psychiatry. 2021. PMID: 33990769
-
Downregulated AKT-mTOR signaling pathway proteins in dorsolateral prefrontal cortex in Schizophrenia.Neuropsychopharmacology. 2020 May;45(6):1059-1067. doi: 10.1038/s41386-020-0614-2. Epub 2020 Jan 17. Neuropsychopharmacology. 2020. PMID: 31952070 Free PMC article.
-
mRNP assembly, axonal transport, and local translation in neurodegenerative diseases.Brain Res. 2018 Aug 15;1693(Pt A):75-91. doi: 10.1016/j.brainres.2018.02.018. Epub 2018 Feb 17. Brain Res. 2018. PMID: 29462608 Free PMC article. Review.
References
-
- Gordon-Weeks P. R. (2005) Developmental and Cell Biology Series (No. 37), Cambridge University Press, Cambridge, UK
-
- Brewer G. J., Cotman C. W. (1989) NMDA receptor regulation of neuronal morphology in cultured hippocampal neurons. Neurosci. Lett. 99, 268–273 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

