The sympathetic nervous system is controlled by transient receptor potential vanilloid 1 in the regulation of body temperature
- PMID: 26136480
- PMCID: PMC4650996
- DOI: 10.1096/fj.15-272526
The sympathetic nervous system is controlled by transient receptor potential vanilloid 1 in the regulation of body temperature
Abstract
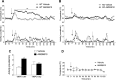
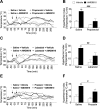
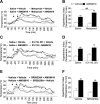
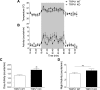
Transient receptor potential vanilloid 1 (TRPV1) is involved in sensory nerve nociceptive signaling. Recently, it has been discovered that TRPV1 receptors also regulate basal body temperature in multiple species from mice to humans. In the present study, we investigated whether TRPV1 modulates basal sympathetic nervous system (SNS) activity. C57BL6/J wild-type (WT) mice and TRPV1 knockout (KO) mice were implanted with radiotelemetry probes for measurement of core body temperature. AMG9810 (50 mg/kg) or vehicle (2% DMSO/5% Tween 80/10 ml/kg saline) was injected intraperitoneally. Adrenoceptor antagonists or vehicle (5 ml/kg saline) was injected subcutaneously. In WT mice, the TRPV1 antagonist, AMG9810, caused significant hyperthermia, associated with increased noradrenaline concentrations in brown adipose tissue. The hyperthermia was significantly attenuated by the β-adrenoceptor antagonist propranolol, the mixed α-/β-adrenoceptor antagonist labetalol, and the α1-adrenoceptor antagonist prazosin. TRPV1 KO mice have a normal basal body temperature, indicative of developmental compensation. d-Amphetamine (potent sympathomimetic) caused hyperthermia in WT mice, which was reduced in TRPV1 KO mice, suggesting a decreased sympathetic drive in KOs. This study provides new evidence that TRPV1 controls thermoregulation upstream of the SNS, providing a potential therapeutic target for sympathetic hyperactivity thermoregulatory disorders.
Keywords: TRPV1; brown adipose tissue; thermogenesis.
© FASEB.
Figures




References
-
- Caterina M. J., Schumacher M. A., Tominaga M., Rosen T. A., Levine J. D., Julius D. (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389, 816–824 - PubMed
-
- Tominaga M., Caterina M. J., Malmberg A. B., Rosen T. A., Gilbert H., Skinner K., Raumann B. E., Basbaum A. I., Julius D. (1998) The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron 21, 531–543 - PubMed
-
- Gavva N. R., Bannon A. W., Surapaneni S., Hovland D. N. Jr, Lehto S. G., Gore A., Juan T., Deng H., Han B., Klionsky L., Kuang R., Le A., Tamir R., Wang J., Youngblood B., Zhu D., Norman M. H., Magal E., Treanor J. J., Louis J. C. (2007) The vanilloid receptor TRPV1 is tonically activated in vivo and involved in body temperature regulation. J. Neurosci. 27, 3366–3374 - PMC - PubMed
-
- Gavva N. R., Treanor J. J., Garami A., Fang L., Surapaneni S., Akrami A., Alvarez F., Bak A., Darling M., Gore A., Jang G. R., Kesslak J. P., Ni L., Norman M. H., Palluconi G., Rose M. J., Salfi M., Tan E., Romanovsky A. A., Banfield C., Davar G. (2008) Pharmacological blockade of the vanilloid receptor TRPV1 elicits marked hyperthermia in humans. Pain 136, 202–210 - PubMed
-
- Yamashita H., Wang Z., Wang Y., Furuyama T., Kontani Y., Sato Y., Mori N. (2008) Impaired basal thermal homeostasis in rats lacking capsaicin-sensitive peripheral small sensory neurons. J. Biochem. 143, 385–393 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- BHF_/British Heart Foundation/United Kingdom
- BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- 20264/VAC_/Versus Arthritis/United Kingdom
- 19296/ARC_/Arthritis Research UK/United Kingdom
- NC/K001531/1/NC3RS_/National Centre for the Replacement, Refinement and Reduction of Animals in Research/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

