Molecular and cellular development of spinal cord locomotor circuitry
- PMID: 26136656
- PMCID: PMC4468382
- DOI: 10.3389/fnmol.2015.00025
Molecular and cellular development of spinal cord locomotor circuitry
Abstract
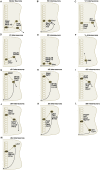
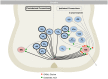
The spinal cord of vertebrate animals is comprised of intrinsic circuits that are capable of sensing the environment and generating complex motor behaviors. There are two major perspectives for understanding the biology of this complicated structure. The first approaches the spinal cord from the point of view of function and is based on classic and ongoing research in electrophysiology, adult behavior, and spinal cord injury. The second view considers the spinal cord from a developmental perspective and is founded mostly on gene expression and gain-of-function and loss-of-function genetic experiments. Together these studies have uncovered functional classes of neurons and their lineage relationships. In this review, we summarize our knowledge of developmental classes, with an eye toward understanding the functional roles of each group.
Keywords: circuit; interneuron; locomotion; motor neuron; sensory; transcription factor.
Figures



References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

