Intravital fluorescence imaging of mouse brain using implantable semiconductor devices and epi-illumination of biological tissue
- PMID: 26137364
- PMCID: PMC4467724
- DOI: 10.1364/BOE.6.001553
Intravital fluorescence imaging of mouse brain using implantable semiconductor devices and epi-illumination of biological tissue
Abstract
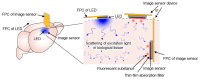
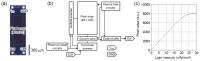
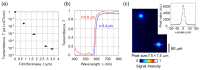
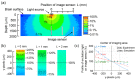
The application of the fluorescence imaging method to living animals, together with the use of genetically engineered animals and synthesized photo-responsive compounds, is a powerful method for investigating brain functions. Here, we report a fluorescence imaging method for the brain surface and deep brain tissue that uses compact and mass-producible semiconductor imaging devices based on complementary metal-oxide semiconductor (CMOS) technology. An image sensor chip was designed to be inserted into brain tissue, and its size was 1500 × 450 μm. Sample illumination is also a key issue for intravital fluorescence imaging. Hence, for the uniform illumination of the imaging area, we propose a new method involving the epi-illumination of living biological tissues, and we performed investigations using optical simulations and experimental evaluation.
Keywords: (110.2970) Image detection systems; (170.2655) Functional monitoring and imaging; (280.1415) Biological sensing and sensors.
Figures


Similar articles
-
Neural Monitoring With CMOS Image Sensors.Basic Clin Neurosci. 2018 May-Jun;9(3):227-235. doi: 10.29252/NIRP.BCN.9.3.227. Basic Clin Neurosci. 2018. PMID: 30034653 Free PMC article.
-
Real-Time Sensing with Patterned Plasmonic Substrates and a Compact Imager Chip.Methods Mol Biol. 2019;2027:87-100. doi: 10.1007/978-1-4939-9616-2_8. Methods Mol Biol. 2019. PMID: 31309475
-
Novel implantable imaging system for enabling simultaneous multiplanar and multipoint analysis for fluorescence potentiometry in the visual cortex.Biosens Bioelectron. 2012 Oct-Dec;38(1):321-30. doi: 10.1016/j.bios.2012.06.035. Epub 2012 Jun 29. Biosens Bioelectron. 2012. PMID: 22784497
-
Microhotplates for Metal Oxide Semiconductor Gas Sensor Applications-Towards the CMOS-MEMS Monolithic Approach.Micromachines (Basel). 2018 Oct 29;9(11):557. doi: 10.3390/mi9110557. Micromachines (Basel). 2018. PMID: 30715056 Free PMC article. Review.
-
A review of recent progress in lens-free imaging and sensing.Biosens Bioelectron. 2017 Feb 15;88:130-143. doi: 10.1016/j.bios.2016.07.115. Epub 2016 Aug 1. Biosens Bioelectron. 2017. PMID: 27503410 Review.
Cited by
-
Self-Reset Image Sensor With a Signal-to-Noise Ratio Over 70 dB and Its Application to Brain Surface Imaging.Front Neurosci. 2021 Jun 15;15:667932. doi: 10.3389/fnins.2021.667932. eCollection 2021. Front Neurosci. 2021. PMID: 34211365 Free PMC article.
-
Thin and Scalable Hybrid Emission Filter via Plasma Etching for Low-Invasive Fluorescence Detection.Sensors (Basel). 2023 Apr 3;23(7):3695. doi: 10.3390/s23073695. Sensors (Basel). 2023. PMID: 37050755 Free PMC article.
-
Multi-Region Microdialysis Imaging Platform Revealed Dorsal Raphe Nucleus Calcium Signaling and Serotonin Dynamics during Nociceptive Pain.Int J Mol Sci. 2023 Apr 3;24(7):6654. doi: 10.3390/ijms24076654. Int J Mol Sci. 2023. PMID: 37047627 Free PMC article.
-
New Bioengineering Breakthroughs and Enabling Tools in Regenerative Medicine.Curr Stem Cell Rep. 2017;3(2):83-97. doi: 10.1007/s40778-017-0081-9. Epub 2017 May 4. Curr Stem Cell Rep. 2017. PMID: 28596936 Free PMC article. Review.
References
-
- Mank M., Santos A. F., Direnberger S., Mrsic-Flogel T. D., Hofer S. B., Stein V., Hendel T., Reiff D. F., Levelt C., Borst A., Bonhoeffer T., Hübener M., Griesbeck O., “A genetically encoded calcium indicator for chronic in vivo two-photon imaging,” Nat. Methods 5(9), 805–811 (2008).10.1038/nmeth.1243 - DOI - PubMed
-
- Wallace D. J., Meyer zum Alten Borgloh S., Astori S., Yang Y., Bausen M., Kügler S., Palmer A. E., Tsien R. Y., Sprengel R., Kerr J. N., Denk W., Hasan M. T., “Single-spike detection in vitro and in vivo with a genetic Ca2+ sensor,” Nat. Methods 5(9), 797–804 (2008).10.1038/nmeth.1242 - DOI - PubMed
LinkOut - more resources
Full Text Sources
