Platelet Inhibitors Reduce Rupture in a Mouse Model of Established Abdominal Aortic Aneurysm
- PMID: 26139462
- PMCID: PMC4552620
- DOI: 10.1161/ATVBAHA.115.305537
Platelet Inhibitors Reduce Rupture in a Mouse Model of Established Abdominal Aortic Aneurysm
Erratum in
-
Correction.Arterioscler Thromb Vasc Biol. 2016 Feb;36(2):e18. doi: 10.1161/ATV.0000000000000030. Arterioscler Thromb Vasc Biol. 2016. PMID: 26819468 No abstract available.
Abstract
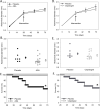
Objective: Rupture of abdominal aortic aneurysms causes a high morbidity and mortality in the elderly population. Platelet-rich thrombi form on the surface of aneurysms and may contribute to disease progression. In this study, we used a pharmacological approach to examine a role of platelets in established aneurysms induced by angiotensin II infusion into hypercholesterolemic mice.
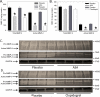
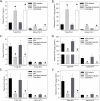
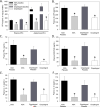
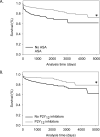
Approach and results: Administration of the platelet inhibitors aspirin or clopidogrel bisulfate to established abdominal aortic aneurysms dramatically reduced rupture. These platelet inhibitors reduced abdominal aortic platelet and macrophage recruitment resulting in decreased active matrix metalloproteinase-2 and matrix metalloproteinase-9. Platelet inhibitors also resulted in reduced plasma concentrations of platelet factor 4, cytokines, and components of the plasminogen activation system in mice. To determine the validity of these findings in human subjects, a cohort of aneurysm patients were retrospectively analyzed using developed and validated algorithms in the electronic medical record database at Vanderbilt University. Similar to mice, administration of aspirin or P2Y12 inhibitors was associated with reduced death among patients with abdominal aortic aneurysm.
Conclusions: These results suggest that platelets contribute to abdominal aortic aneurysm progression and rupture.
Keywords: angiotensin II; aortic aneurysm, abdominal; aspirin; blood platelets; clopidogrel; mice.
© 2015 American Heart Association, Inc.
Figures


Similar articles
-
CD40L Deficiency Protects Against Aneurysm Formation.Arterioscler Thromb Vasc Biol. 2018 May;38(5):1076-1085. doi: 10.1161/ATVBAHA.117.310640. Epub 2018 Mar 8. Arterioscler Thromb Vasc Biol. 2018. PMID: 29519940
-
A Modified Murine Abdominal Aortic Aneurysm Rupture Model Using Elastase Perfusion and Angiotensin II Infusion.Ann Vasc Surg. 2020 Aug;67:474-481. doi: 10.1016/j.avsg.2020.03.002. Epub 2020 Mar 12. Ann Vasc Surg. 2020. PMID: 32171859
-
Ginsenoside Rb1 attenuates angiotensin II-induced abdominal aortic aneurysm through inactivation of the JNK and p38 signaling pathways.Vascul Pharmacol. 2015 Oct;73:86-95. doi: 10.1016/j.vph.2015.04.003. Epub 2015 Apr 22. Vascul Pharmacol. 2015. PMID: 25912763
-
Natural regulatory T cells limit angiotensin II-induced aneurysm formation and rupture in mice.Arterioscler Thromb Vasc Biol. 2013 Oct;33(10):2374-9. doi: 10.1161/ATVBAHA.113.301280. Epub 2013 Aug 1. Arterioscler Thromb Vasc Biol. 2013. PMID: 23908246
-
Antithrombotic therapy in abdominal aortic aneurysm: beneficial or detrimental?Blood. 2018 Dec 20;132(25):2619-2628. doi: 10.1182/blood-2017-08-743237. Epub 2018 Sep 18. Blood. 2018. PMID: 30228233 Free PMC article. Review.
Cited by
-
Two C-C Family Chemokines, Eotaxin and RANTES, Are Novel Independent Plasma Biomarkers for Abdominal Aortic Aneurysm.J Am Heart Assoc. 2016 Apr 28;5(5):e002993. doi: 10.1161/JAHA.115.002993. J Am Heart Assoc. 2016. PMID: 27126477 Free PMC article.
-
The impact of endovascular stents types on perioperative outcomes of ruptured abdominal aortic aneurysms: a single-center experience.Front Cardiovasc Med. 2024 Mar 13;11:1272389. doi: 10.3389/fcvm.2024.1272389. eCollection 2024. Front Cardiovasc Med. 2024. PMID: 38545342 Free PMC article.
-
Imaging Techniques for Aortic Aneurysms and Dissections in Mice: Comparisons of Ex Vivo, In Situ, and Ultrasound Approaches.Biomolecules. 2022 Feb 21;12(2):339. doi: 10.3390/biom12020339. Biomolecules. 2022. PMID: 35204838 Free PMC article. Review.
-
Molecular pathogenesis of genetic and sporadic aortic aneurysms and dissections.Curr Probl Surg. 2017 Mar;54(3):95-155. doi: 10.1067/j.cpsurg.2017.01.001. Epub 2017 Feb 3. Curr Probl Surg. 2017. PMID: 28521856 Free PMC article. Review. No abstract available.
-
Abdominal aortic aneurysms and platelets: infiltration, inflammation, and elastin disintegration.Cardiovasc Res. 2024 Mar 30;120(4):331-332. doi: 10.1093/cvr/cvae010. Cardiovasc Res. 2024. PMID: 38193624 Free PMC article. No abstract available.
References
-
- Alcorn HG, Wolfson SK, Jr., Sutton-Tyrrell K, Kuller LH, O'Leary D. Risk factors for abdominal aortic aneurysms in older adults enrolled in The Cardiovascular Health Study. Arterioscler Thromb Vasc Biol. 1996;16:963–970. - PubMed
-
- Bengtsson H, Bergqvist D, Sternby NH. Increasing prevalence of abdominal aortic aneurysms. A necropsy study. Eur J Surg. 1992;158:19–23. - PubMed
-
- Daugherty A, Cassis LA. Mouse models of abdominal aortic aneurysms. Arterioscler Thromb Vasc Biol. 2004;24:429–434. - PubMed
-
- Saraff K, Babamusta F, Cassis LA, Daugherty A. Aortic dissection precedes formation of aneurysms and atherosclerosis in angiotensin II-infused, apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol. 2003;23:1621–1626. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

